Introduction to Chromophores
Chromophores are fundamental chemical groups responsible for the absorption of light in the ultraviolet (UV) and visible regions of the electromagnetic spectrum. They play a crucial role in determining the color of compounds and are extensively studied in UV-Visible spectroscopy, medicinal chemistry, photochemistry, and dye chemistry.
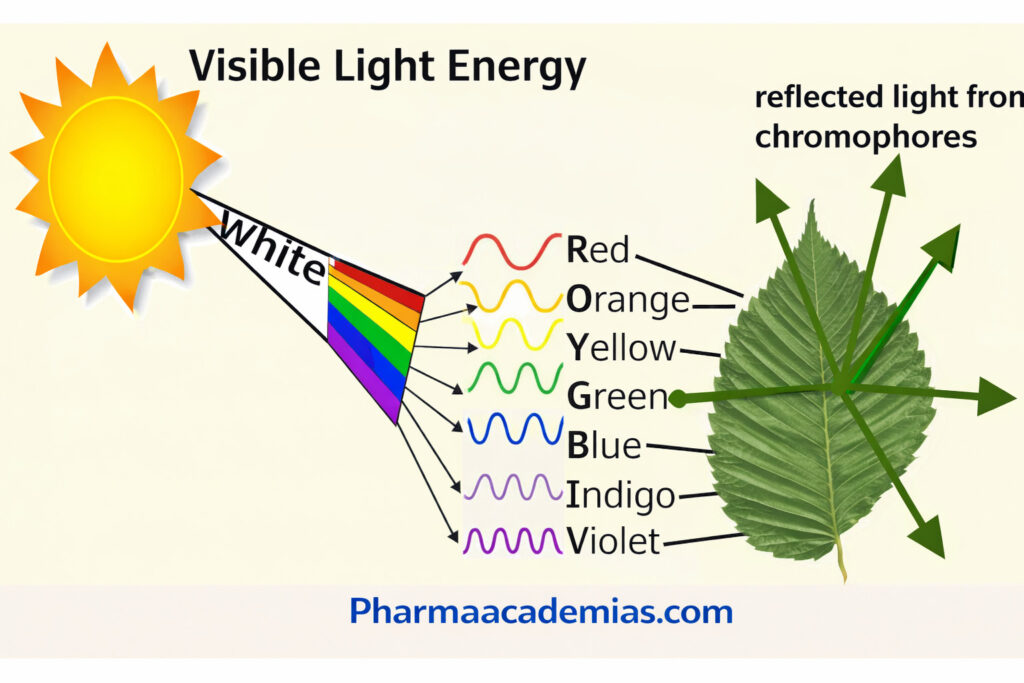
The term “chromophore” is derived from Greek words “chroma” (color) and “phoros” (bearing), meaning “color-bearing group.” These groups absorb specific wavelengths of light, leading to electronic transitions within molecules, which ultimately result in color perception.
Definition of Chromophores
A chromophore is a part of a molecule, typically consisting of conjugated π-electron systems, that absorbs light in the UV or visible region and is responsible for the color of the compound.
In simple terms, chromophores are light-absorbing functional groups that cause electronic excitation when exposed to radiation.
Basic Principle of Chromophore Action
When light interacts with a molecule containing a chromophore, electrons in the molecule absorb energy and transition from a lower energy state (ground state) to a higher energy state (excited state).
This absorption typically involves:
- π → π* transitions (common in double bonds and aromatic systems)
- n → π* transitions (involving lone pair electrons, e.g., in carbonyl groups)
The specific wavelength absorbed depends on the structure and conjugation of the chromophore.
Characteristics of Chromophores
Chromophores exhibit distinct physicochemical and spectroscopic properties that influence their behavior:
- They generally contain unsaturated groups such as double bonds or aromatic rings, which allow electron delocalization.
- The presence of conjugation significantly increases the wavelength of absorption (bathochromic shift), often shifting absorption from UV to visible region.
- Chromophores display specific absorption maxima (λmax), which is a key parameter in UV-Visible spectroscopy.
- Their absorption intensity is expressed in terms of molar absorptivity (ε), which indicates how strongly a substance absorbs light.
- The color observed is complementary to the wavelength absorbed, meaning if a chromophore absorbs blue light, it may appear orange.
Types of Chromophores
Chromophores can be classified based on their structure and electronic transitions.
Simple Chromophores
Simple chromophores contain isolated unsaturated groups without extensive conjugation. These typically absorb in the UV region.
Examples include:
- Carbonyl group (C=O)
- Nitroso group (–NO)
- Nitro group (–NO₂)
These groups show relatively weak absorption and are often colorless.
Conjugated Chromophores
Conjugated chromophores contain alternating double and single bonds, allowing delocalization of π-electrons.
Examples include:
- Conjugated dienes
- Polyenes
- Aromatic rings
As conjugation increases, the absorption shifts toward longer wavelengths, often entering the visible region, producing color.
Auxochrome-Modified Chromophores
Auxochromes are groups that themselves do not act as chromophores but enhance the intensity and wavelength of absorption when attached to a chromophore.
Common auxochromes include:
- –OH (hydroxyl)
- –NH₂ (amino)
- –OR (alkoxy)
They cause shifts such as:
- Bathochromic shift (red shift) → increase in wavelength
- Hyperchromic effect → increase in absorption intensity
Aromatic Chromophores
Aromatic compounds such as benzene and its derivatives act as chromophores due to their conjugated π-electron systems.
Substituted aromatic compounds exhibit strong absorption and are widely used in dyes and pharmaceuticals.
Extended Chromophoric Systems
These include large conjugated systems such as carotenoids and synthetic dyes, which absorb visible light strongly and exhibit intense colors.
Important Spectral Shifts in Chromophores
Chromophores exhibit characteristic shifts in absorption under different conditions:
- Bathochromic shift (Red shift): Shift to longer wavelength due to increased conjugation or solvent effects
- Hypsochromic shift (Blue shift): Shift to shorter wavelength due to reduced conjugation
- Hyperchromic effect: Increase in absorption intensity
- Hypochromic effect: Decrease in absorption intensity
These shifts are crucial in interpreting UV-Visible spectra.
Applications of Chromophores
Chromophores have wide-ranging applications across various scientific and industrial fields:
1. UV-Visible Spectroscopy
Chromophores are the basis of UV-Visible spectroscopic analysis, allowing identification and quantification of compounds based on their absorption spectra.
2. Pharmaceutical Analysis
They are used in drug analysis for:
- Determination of drug concentration
- Stability studies
- Detection of impurities
Many drugs contain chromophoric groups that enable spectrophotometric estimation.
3. Dye and Pigment Industry
Chromophores are responsible for the color of dyes and pigments used in textiles, inks, and paints. By modifying chromophores and auxochromes, a wide range of colors can be produced.
4. Biological Systems
In biological molecules, chromophores play essential roles:
- Hemoglobin contains a heme chromophore responsible for oxygen transport
- Chlorophyll absorbs light for photosynthesis
- Visual pigments in the eye rely on chromophoric systems
5. Photochemistry and Photobiology
Chromophores absorb light energy and initiate photochemical reactions, which are important in:
- Photodynamic therapy
- Solar energy conversion
- Photostability studies
6. Environmental and Analytical Chemistry
Chromophores are used for detecting pollutants and analyzing water quality through spectroscopic methods.
Conclusion
Chromophores are essential structural components that determine the light-absorbing properties and color of molecules. Their behavior is governed by electronic transitions and molecular structure, particularly conjugation. Understanding chromophores is fundamental to spectroscopy, pharmaceutical analysis, and many industrial applications. With advancements in material science and photochemistry, chromophores continue to play a critical role in modern science and technology.





