Gas Chromatography (GC) is a highly efficient analytical separation technique used for the analysis of volatile and semi-volatile compounds present in complex mixtures. It is one of the most widely used chromatographic techniques in pharmaceutical analysis, environmental monitoring, forensic toxicology, petrochemical industries, and food quality control.
The fundamental idea behind GC is the separation of components based on their distribution between a mobile gaseous phase (carrier gas) and a stationary phase (liquid or solid coated inside a column). The components of a sample travel through the column at different speeds depending on their volatility and interaction with the stationary phase, leading to separation.
GC is especially valuable because it provides high sensitivity, excellent resolution, and fast analysis time, making it ideal for trace-level detection of compounds.
Theory of Gas Chromatography
The theory of Gas Chromatography is based on the principle of partitioning and adsorption of analyte molecules between two phases: the mobile phase (inert gas) and the stationary phase.
When a sample is injected into the heated injector, it is rapidly vaporized and carried by the inert gas through the column. Inside the column, repeated adsorption and desorption occur between the analyte molecules and the stationary phase. This continuous equilibrium process causes separation of components.
Also read this
Mechanism of Separation
Each compound distributes itself between the gas phase and stationary phase according to its:
- Volatility (boiling point)
- Polarity
- Molecular interactions (van der Waals forces, dipole interactions, hydrogen bonding)
- Partition coefficient
Compounds with higher volatility and lower interaction with stationary phase move faster and elute earlier. Conversely, compounds with strong interaction or higher boiling point move slowly and elute later.
The separation efficiency is influenced by:
- Column temperature
- Nature of stationary phase
- Flow rate of carrier gas
- Column length and diameter
The elution of compounds is represented in the form of a chromatogram, where each peak corresponds to a different component, and retention time is used for identification.
Instrumentation of Gas Chromatography
Gas Chromatography consists of several interconnected components, each playing a crucial role in achieving efficient separation and detection.
1. Carrier Gas System
The carrier gas acts as the mobile phase and transports the vaporized sample through the column. It must be chemically inert to avoid reactions with analytes or stationary phase.
Common carrier gases include helium, nitrogen, and hydrogen. Helium is widely preferred due to its optimal efficiency and safety balance. Hydrogen provides faster analysis but is flammable, while nitrogen is economical but less efficient at high flow rates.
The flow of carrier gas is regulated using pressure controllers and flow meters to ensure reproducibility.
2. Sample Injection System
The injection system introduces the sample into the chromatographic system. It is usually a heated injection port maintained at a temperature higher than the boiling point of analytes to ensure instant vaporization.
Samples are introduced using microsyringes or autosamplers. The injector is designed to avoid sample decomposition and ensure complete volatilization. Split and splitless injection techniques are commonly used:
- Split injection: Only a small portion enters the column (used for concentrated samples)
- Splitless injection: Entire sample enters the column (used for trace analysis)
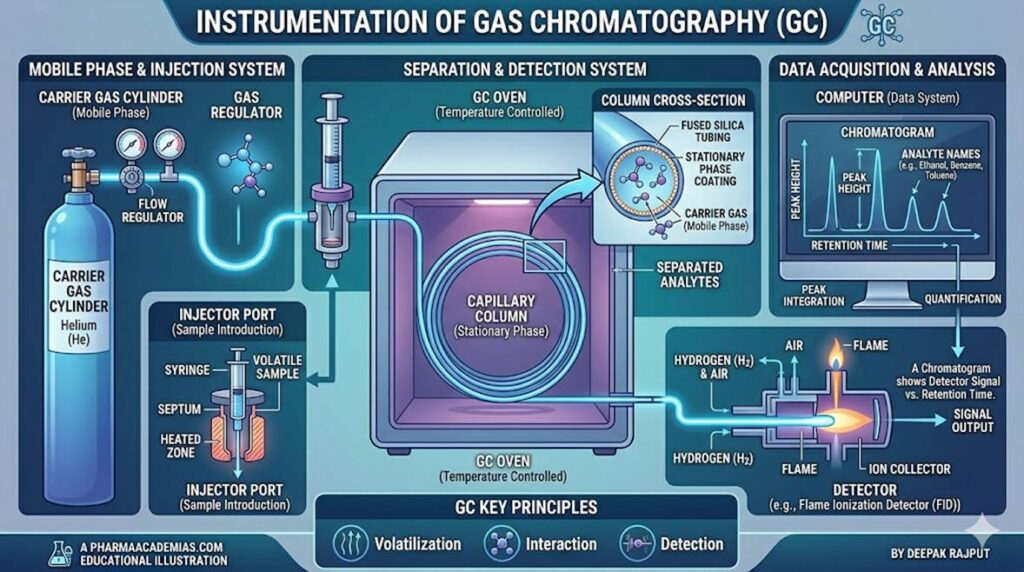
3. Chromatographic Column
The column is the heart of GC where actual separation occurs. There are two main types:
Packed columns contain solid support material coated with stationary phase, while capillary columns (open tubular columns) consist of a thin film of stationary phase coated on the inner wall of a fused silica tube. Capillary columns offer higher efficiency, better resolution, and faster analysis, and are widely used in modern GC systems.
Column length, diameter, and film thickness significantly influence separation performance.
4. Stationary Phase
The stationary phase is a non-volatile liquid or solid that interacts with analytes during separation. Its polarity determines selectivity.
Non-polar stationary phases (e.g., polydimethylsiloxane) separate compounds mainly based on volatility, while polar stationary phases (e.g., polyethylene glycol) provide separation based on polarity differences.
5. Column Oven
The column is housed inside a temperature-controlled oven. The oven maintains precise temperature conditions and allows temperature programming to optimize separation of complex mixtures.
6. Detectors
Detectors convert chemical information into an electrical signal, producing a chromatogram.
Common detectors include:
The Flame Ionization Detector (FID) is widely used due to its high sensitivity for organic compounds. It works by burning analytes in a hydrogen-air flame and measuring ion formation.
The Thermal Conductivity Detector (TCD) measures changes in thermal conductivity of the gas stream and is universal but less sensitive.
The Electron Capture Detector (ECD) is highly sensitive to electronegative compounds such as halogenated substances and is widely used in pesticide analysis.
The Mass Spectrometer (GC-MS) provides both separation and structural identification, making it the most powerful GC detection system.
7. Recording System
The output from the detector is recorded as a chromatogram, which displays peaks representing individual components. The position of peaks indicates retention time, while peak area represents concentration.
Derivatization in Gas Chromatography
Derivatization is a chemical modification process used to enhance the volatility, stability, and detectability of analytes that are otherwise unsuitable for GC analysis.
Many biologically important compounds such as amino acids, sugars, fatty acids, and steroids are polar and thermally unstable. These compounds are chemically transformed into volatile derivatives.
Purpose of Derivatization
- Increases volatility
- Reduces polarity
- Improves thermal stability
- Enhances detector response
- Improves peak shape and resolution
Common Derivatization Methods
Silylation is the most widely used method where active hydrogen atoms are replaced with trimethylsilyl groups, making compounds more volatile and stable.
Acylation introduces acyl groups, reducing hydrogen bonding and increasing volatility.
Alkylation converts polar functional groups into esters or ethers, improving chromatographic behavior.
Temperature Programming in Gas Chromatography
Temperature programming is the controlled variation of column temperature during a chromatographic run to improve separation efficiency.
Instead of maintaining a constant temperature, the column temperature is gradually increased according to a predefined program.
At lower temperatures, volatile compounds separate efficiently, while at higher temperatures, less volatile compounds are eluted more quickly, reducing analysis time and preventing peak broadening.
Advantages of Temperature Programming
It improves resolution for complex mixtures containing compounds with a wide boiling point range. It also reduces total analysis time and enhances peak symmetry, especially for late-eluting compounds.
Advantages of Gas Chromatography
Gas Chromatography offers several significant advantages. It provides excellent separation efficiency with high resolution, even for complex mixtures. It requires very small sample volumes and offers rapid analysis compared to other chromatographic methods.
GC is highly sensitive and capable of detecting compounds at trace levels. It also provides excellent reproducibility, making it suitable for both qualitative and quantitative analysis.
When coupled with mass spectrometry, GC becomes an extremely powerful analytical tool capable of identifying unknown compounds with high accuracy.
Disadvantages of Gas Chromatography
Despite its advantages, GC has several limitations. It is restricted to volatile and thermally stable compounds, meaning non-volatile or thermolabile compounds cannot be analyzed directly.
Derivatization may be required for certain compounds, which increases sample preparation time and complexity. GC instruments are relatively expensive and require skilled operation and maintenance.
Additionally, contamination of carrier gas or column degradation can affect accuracy and performance.
Applications of Gas Chromatography
Gas Chromatography has extensive applications across multiple fields.
In pharmaceutical analysis, it is used for residual solvent analysis, purity testing, and quality control of drug substances. In environmental analysis, GC is used to detect volatile organic compounds, pesticides, and pollutants in air and water samples.
In forensic science, it plays a critical role in detecting alcohol, drugs of abuse, and toxic substances. In the food industry, GC is used for flavor profiling, fragrance analysis, and detection of food contaminants and additives.
In petrochemical industries, GC is essential for analyzing crude oil components, hydrocarbons, natural gas composition, and fuel quality.
It is also used in clinical and biomedical research for metabolic profiling and biomarker identification.
Conclusion
Gas Chromatography is a highly efficient and versatile analytical technique used for the separation, identification, and quantification of volatile compounds. Its high sensitivity, precision, and wide range of applications make it indispensable in pharmaceutical, environmental, forensic, and industrial analysis.
Although it is limited to volatile and thermally stable compounds, advancements such as derivatization techniques and GC-MS coupling have significantly expanded its applicability. GC remains one of the most important tools in modern analytical chemistry.






