Pilot plant scale-up is a crucial phase in pharmaceutical development that ensures successful transition from laboratory-scale formulations to large-scale manufacturing. Each dosage form—solid, liquid oral, and semi-solid—has unique formulation and processing challenges that must be addressed during scale-up. Along with technical considerations, proper documentation is essential to maintain quality, consistency, and regulatory compliance.
Pilot Plant Scale Up Considerations
Scale-Up Considerations for Solid Dosage Forms
Solid dosage forms such as tablets and capsules are the most commonly manufactured pharmaceutical products. Their scale-up involves multiple unit operations including blending, granulation, drying, compression, and coating.
During scale-up, the uniform mixing of powders is a major concern. Differences in particle size, density, and flow properties can lead to segregation, affecting content uniformity. The choice of mixing equipment and optimization of mixing time are critical to ensure homogeneity.
Granulation, whether wet or dry, must be carefully controlled. Parameters such as binder concentration, granulating fluid addition rate, and mixing speed influence granule size and strength. Scaling up granulation often leads to variations in moisture distribution and granule characteristics.
Drying is another critical step where temperature, airflow, and drying time must be optimized to achieve uniform moisture content without degrading the drug. Overdrying can cause poor compressibility, while underdrying can lead to stability issues.
Compression of tablets requires attention to machine speed, compression force, and tooling. Changes in compression parameters during scale-up may affect hardness, friability, disintegration time, and dissolution profile.
Coating processes, if involved, require optimization of spray rate, inlet air temperature, and coating pan speed to ensure uniform coating without defects such as peeling or cracking.
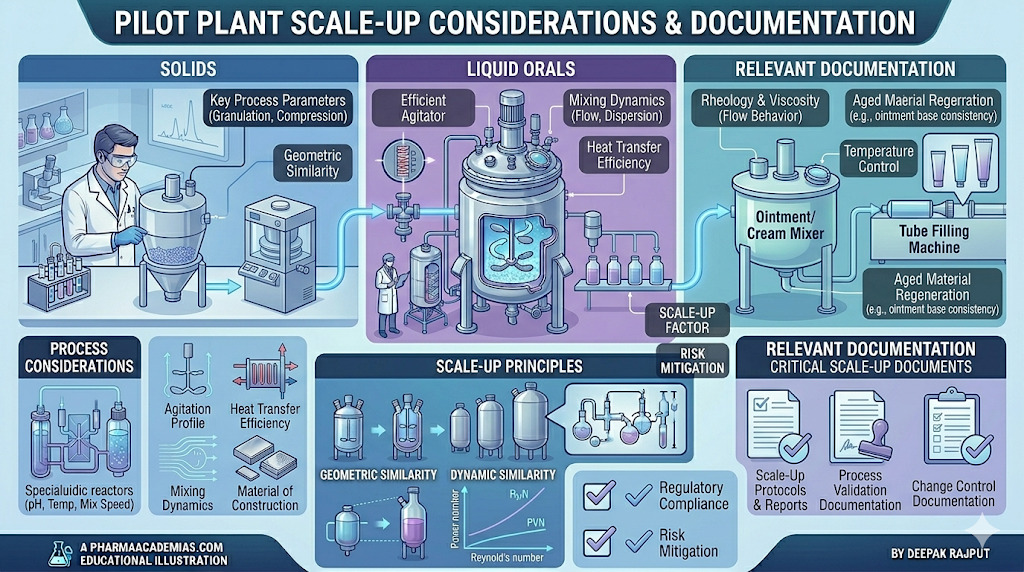
Pilot Scale Up Considerations for Liquid Oral Dosage Forms
Liquid oral formulations include solutions, syrups, suspensions, and emulsions. Their scale-up focuses on achieving uniformity, stability, and microbial safety.
In solutions and syrups, solubility and dissolution rate of active ingredients must be maintained. Mixing efficiency becomes critical, as inadequate mixing may result in concentration gradients.
For suspensions, maintaining uniform dispersion of particles is a major challenge. Sedimentation rate depends on particle size, viscosity, and density differences. Scale-up must ensure proper agitation and use of suspending agents to maintain stability.
In emulsions, droplet size distribution is crucial for stability. The scale-up process must maintain proper shear conditions during emulsification to avoid coalescence or phase separation.
Viscosity control is important for all liquid orals, as it affects pourability, dosing accuracy, and patient acceptability. Additionally, microbial contamination must be controlled by using preservatives and maintaining hygienic manufacturing conditions.
Pilot plant Scale Up Considerations for Semi-Solid Dosage Forms
Semi-solid dosage forms include ointments, creams, gels, and pastes. These formulations require careful control of rheological properties and homogeneity.
Mixing and homogenization are critical steps in semi-solid scale-up. Uniform distribution of active ingredients must be ensured to avoid dose variability. The type of mixer and mixing speed significantly influence product consistency.
Temperature control is essential, especially for formulations containing waxes or thermolabile components. Improper temperature management may lead to phase separation or degradation of ingredients.
Viscosity and spreadability are important quality attributes. Changes in scale may affect shear forces, leading to variations in texture and consistency.
In emulsified semi-solids such as creams, maintaining emulsion stability is crucial. Phase inversion, creaming, or breaking of emulsion can occur if process parameters are not optimized.
Filling and packaging operations also require attention, as semi-solids must be filled uniformly without air entrapment.
Relevant Documentation in Pilot Plant Scale-Up
Documentation is an integral part of pilot plant scale-up, ensuring traceability, reproducibility, and compliance with regulatory standards such as Good Manufacturing Practices (GMP).
The Master Formula Record (MFR) is a key document that provides detailed instructions for manufacturing, including formulation composition, equipment, processing steps, and in-process controls. It serves as a reference for all production batches.
The Batch Manufacturing Record (BMR) is a real-time record of each batch produced. It includes details of raw materials used, process parameters, in-process observations, and final product results. It ensures that each batch is manufactured according to the predefined procedure.
Standard Operating Procedures (SOPs) define step-by-step instructions for performing specific operations such as equipment cleaning, calibration, and processing steps. They ensure consistency and minimize errors.
Validation documents, including process validation protocols and reports, confirm that the manufacturing process consistently produces products meeting quality specifications.
Quality control records document analytical testing of raw materials, in-process samples, and finished products. Stability study reports provide information on shelf life and storage conditions.
Equipment qualification documents, such as Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ), ensure that equipment functions correctly and consistently.
Proper documentation not only ensures regulatory compliance but also facilitates troubleshooting and continuous process improvement.
Conclusion
Pilot plant scale-up for different dosage forms requires a comprehensive understanding of formulation characteristics, process parameters, and equipment behavior. Solid, liquid oral, and semi-solid formulations each present unique challenges that must be carefully managed to ensure product quality and consistency.
Equally important is the maintenance of thorough and accurate documentation, which supports regulatory compliance and ensures reproducibility of the manufacturing process. A well-planned and executed scale-up strategy is essential for successful commercialization of pharmaceutical products.





