The pricing of pharmaceutical products in India is a multi-layered and highly regulated process, designed to balance the interests of the end consumer, pharmaceutical manufacturers, distributors, and the government. The Drugs Price Control Order (DPCO) 2013, issued by the National Pharmaceutical Pricing Authority (NPPA) under the Essential Commodities Act, 1955, provides a comprehensive framework for ensuring that essential medicines remain affordable, accessible, and fairly priced.
Within this framework, three critical price parameters are of central importance:
- Sale price of bulk drugs (API – Active Pharmaceutical Ingredient)
- Retail price of formulations (finished dosage forms)
- Ceiling price of scheduled formulations (maximum price under DPCO 2013)
Understanding the differences, interrelationships, and implications of these prices is essential for pharmaceutical professionals, healthcare policymakers, and regulatory authorities.
1. Sale Price of Bulk Drugs (API Price) DPCO 2013
Definition and Scope
The sale price of bulk drugs is the cost at which active pharmaceutical ingredients (APIs) are sold by manufacturers to pharmaceutical companies or formulators. These APIs are the biologically active substances responsible for the therapeutic effects of drugs.
This price represents the raw material cost, which is the foundation for all subsequent pricing decisions for finished formulations. The sale price of APIs does not include excipients, packaging, taxes, or retail margins—it is purely the cost of the active compound itself.
Determinants of Bulk Drug Prices
The pricing of APIs is influenced by multiple factors, including:
- Raw material availability: Prices of precursors, solvents, or plant extracts used in API synthesis can fluctuate based on supply-demand dynamics and seasonal variations.
- Manufacturing costs: Energy consumption, labor, equipment maintenance, and facility overheads are major cost drivers.
- Technological complexity: APIs that require sophisticated chemical synthesis, multiple-step reactions, or sterile manufacturing processes tend to be more expensive.
- Regulatory compliance: Adherence to Good Manufacturing Practices (GMP), quality assurance protocols, and environmental regulations adds to production costs.
- Global market influences: India is a major supplier of generic APIs internationally. Import-export duties, global demand, and competition with countries like China can affect pricing.
- Economies of scale: Bulk production reduces unit cost; smaller production runs increase per-unit cost.
Importance of Bulk Drug Prices
The sale price of bulk drugs directly affects:
- The cost of finished formulations.
- Profit margins for pharmaceutical manufacturers.
- Accessibility of medicines in the market, especially for low-cost generic drugs.
Example: If the API cost for Metformin 500 mg is ₹800 per kg, then the per-tablet cost contribution from the API is calculated based on the weight of the active ingredient per tablet.
2. Retail Price of Formulations DPCO 2013
The retail price (RP) is the price at which a finished pharmaceutical product—such as tablets, capsules, injections, or syrups—is sold to the end consumer at a pharmacy, hospital, or dispensary. It represents the total cost incurred by the consumer to obtain the medicine.
Components of Retail Price
The retail price is composed of multiple elements:
- API cost: Derived from the bulk drug price as discussed above.
- Excipients: Inactive ingredients such as binders, fillers, stabilizers, and solvents required to formulate the final dosage.
- Manufacturing overheads: Labor, machinery, packaging, quality control, and regulatory compliance costs.
- Taxes: Goods and Services Tax (GST) or other statutory charges levied on pharmaceutical products.
- Distribution costs: Logistics, warehousing, shipping, and handling expenses.
- Margins: Distributor and retailer profit margins, which can vary based on market dynamics.
- Branding and marketing costs: Advertising, doctor promotion, and marketing campaigns.
Market Dynamics
- For non-scheduled drugs, the retail price is largely market-driven and can vary significantly between brands and regions.
- For scheduled drugs, the retail price cannot legally exceed the ceiling price notified by NPPA under DPCO 2013.
Example: A 500 mg Paracetamol tablet may have a retail price of ₹3, consisting of:
- API cost: ₹0.50
- Excipients and packaging: ₹0.50
- Taxes: ₹0.30
- Distributor/retailer margins: ₹1.20
- Marketing/other costs: ₹0.50
The retail price is therefore dynamic, reflecting both cost structure and regulatory limits.
3. Ceiling Price of Scheduled Formulations
The ceiling price is the maximum price at which a scheduled formulation can be sold to the consumer. Scheduled formulations are those included in the National List of Essential Medicines (NLEM, 2011) and regulated under DPCO 2013 to ensure affordability.
Purpose of Ceiling Price
The ceiling price mechanism ensures that:
- Essential drugs remain accessible and affordable to all socioeconomic groups.
- Arbitrary or exploitative pricing by pharmaceutical companies is prevented.
- Healthcare equity is promoted, particularly for chronic and life-saving medications.
- Manufacturers can still make a reasonable profit while maintaining social responsibility.
Calculation of Ceiling Price under DPCO 2013
The calculation follows a market-based pricing methodology:
- Identify all brands of the scheduled drug with a market share of 1% or more.
- Calculate the simple average of these brands’ retail prices.
- Include all applicable taxes to determine the Maximum Retail Price (MRP).
- Publish the ceiling price, which becomes legally enforceable.
Example of Ceiling Price Calculation:
Suppose four brands of Paracetamol 500 mg tablets have MRPs of ₹2.50, ₹3.00, ₹3.20, and ₹2.80.
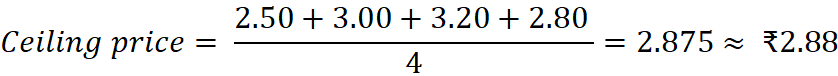
This means no manufacturer or retailer can sell above ₹2.88, ensuring affordability and compliance with DPCO 2013.
Legal and Regulatory Implications
- Violation of ceiling prices can result in:
- Fines
- Confiscation of excess profits
- Legal action under the Essential Commodities Act
- NPPA continuously monitors market prices through:
- Market surveys
- Mandatory reporting by manufacturers
- Audits and inspections
4. Interrelationship Between Bulk Drug Price, Retail Price, and Ceiling Price
| Parameter | Definition | Determined by | Relevance/Impact |
| Sale Price of Bulk Drug | Cost of API sold to manufacturers | Production cost, raw material availability, technological complexity, global supply | Forms the base cost of formulation; influences retail price indirectly |
| Retail Price of Formulation | Price paid by end consumer | API cost, excipients, manufacturing, taxes, margins, marketing | Determines affordability; may vary for non-scheduled drugs |
| Ceiling Price of Scheduled Formulation | Maximum permissible retail price | NPPA calculation based on market average of scheduled formulations | Ensures affordability; legally enforceable; prevents overpricing |
Key Insights
- The bulk drug price is a fundamental determinant of manufacturing cost but does not directly control the retail price.
- The retail price reflects total production costs, taxes, distribution, and profit margins.
- The ceiling price is a regulatory cap, ensuring that the retail price does not exceed the legal limit for scheduled drugs.
- For non-scheduled drugs, retail prices are market-driven and can exceed the average price of other brands.
Check this: QSEM and Q-Series Guidelines –
5. Practical Implications of DPCO 2013 Pricing
For Manufacturers
- Must ensure all scheduled drugs comply with the ceiling price.
- Maintain detailed reports of production, sales, and pricing for NPPA verification.
- Strategically price non-scheduled drugs while adhering to regulated formulations.
For Distributors and Retailers
- Legally obliged to sell scheduled drugs at or below the ceiling price.
- Cannot inflate prices through excessive margins or surcharges.
For Consumers
- Guaranteed access to affordable essential medicines.
- Reduced financial burden for chronic disease management and life-saving therapies.
For Regulators (NPPA)
- Monitors the market and enforces compliance.
- Revises ceiling prices periodically based on inflation, production costs, and market changes.
- Protects public interest while balancing industrial growth.
6. Expanded Example to Illustrate Interconnections
Scenario: Consider Metformin 500 mg tablets:
- API Cost: ₹800/kg → contributes ₹0.60 per tablet
- Excipients and packaging: ₹0.40 per tablet
- Manufacturing overhead: ₹0.30 per tablet
- Distributor margin: ₹0.30
- Retailer margin: ₹0.50
- Taxes (GST): ₹0.30
Total Retail Price: ₹2.40
Now, if NPPA calculates the ceiling price as ₹2.50:
- Retailer and manufacturer cannot sell above ₹2.50.
- API cost fluctuations can be absorbed within the margin or trigger price revision by NPPA.
This ensures that affordable access is maintained without jeopardizing manufacturer sustainability.
Conclusion
- The sale price of bulk drugs sets the foundation for production cost.
- The retail price determines what the consumer pays, incorporating production cost, margins, and taxes.
- The ceiling price of scheduled formulations, as dictated by DPCO 2013, ensures affordability, equity, and legal compliance.
- This multi-layered pricing mechanism reflects a balance between market forces, public health needs, and industrial viability, making DPCO 2013 a landmark regulation in India’s pharmaceutical policy.