UV-Visible Spectroscopy (UV-Vis Spectroscopy) is one of the most widely used analytical techniques in chemistry, biochemistry, pharmaceutical sciences, environmental science, and materials research. It measures the absorption or reflectance of ultraviolet and visible light in the wavelength range of 200–800 nm by atoms, ions, or molecules. The technique is highly valuable for both qualitative identification and quantitative determination of compounds.
This method is especially useful for substances containing chromophores, such as conjugated double bonds, aromatic rings, carbonyl groups, and other functional groups capable of electronic transitions.
Principle of UV Visible Spectroscopy
The fundamental principle of UV-Visible spectroscopy is based on the absorption of electromagnetic radiation by molecules. When UV or visible light passes through a sample, certain wavelengths are absorbed by the substance. The absorbed energy excites electrons from a lower-energy ground state to a higher-energy excited state.
The type of absorption depends on the molecular structure and usually involves:
- π → π* transitions in unsaturated and aromatic compounds
- n → π* transitions in compounds containing lone pair electrons
- σ → σ* transitions in high-energy UV regions
The wavelength at which maximum absorption occurs is known as λmax, and it serves as a characteristic fingerprint for many compounds.
Beer-Lambert Law
The quantitative basis of UV-Visible spectroscopy is the Beer-Lambert Law, which relates absorbance to concentration.
A = εcl
Where:
- A = absorbance
- ε = molar absorptivity or molar extinction coefficient
- c = concentration of the solution
- l = path length of the cuvette
Another important expression is:
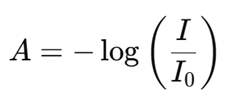
Where I₀ is the intensity of incident light and I is the transmitted light intensity. This law forms the foundation for concentration determination in unknown samples.
Instrumentation of UV Visible Spectrophotometer
A UV-Visible spectrophotometer consists of several important components that work together to produce an absorbance spectrum.
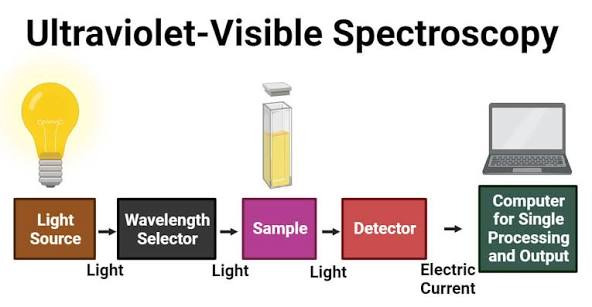
Light Source
The instrument uses separate lamps for different regions of the spectrum. A deuterium lamp is commonly used for the ultraviolet region, while a tungsten-halogen lamp is used for visible light measurements.
Monochromator
The monochromator separates polychromatic light into individual wavelengths using a prism or diffraction grating. It ensures that only the selected wavelength passes through the sample.
Sample Holder
The sample is placed in a cuvette, which is usually made of quartz for UV measurements and glass for visible measurements. Quartz is preferred because it is transparent in the UV region.
Detector
The detector measures the transmitted light intensity after it passes through the sample. Common detectors include photodiodes, photocells, and photomultiplier tubes.
Data Processor and Display
The measured signal is converted into digital form and displayed as an absorbance vs wavelength spectrum for interpretation.
Working Mechanism of UV Visible Spectroscopy
The working process of UV-Visible spectroscopy follows a systematic sequence.
First, the light source emits radiation covering the UV and visible range. The monochromator then isolates a specific wavelength. This monochromatic light passes through the sample solution contained in the cuvette.
If the sample contains molecules capable of absorbing that wavelength, part of the light energy is absorbed while the remaining light is transmitted. The detector measures the transmitted intensity and compares it with the blank or reference solvent. The instrument software then converts this data into absorbance values and plots the absorption spectrum.
The resulting spectrum provides valuable information about the identity, concentration, and purity of the analyte.
Applications of UV Visible Spectroscopy
UV-Visible spectroscopy has broad applications across multiple scientific and industrial fields.
In quantitative analysis, it is widely used to determine the concentration of analytes such as drugs, dyes, metal complexes, and food additives using calibration curves.
In qualitative analysis, the technique helps identify functional groups, chromophores, and conjugation patterns based on λmax values and spectral shifts.
In pharmaceutical analysis, UV-Vis spectroscopy is essential for drug assay, dissolution studies, impurity analysis, and quality control testing.
In biochemistry, it is used for protein estimation at 280 nm and DNA/RNA quantification at 260 nm, making it indispensable in molecular biology laboratories.
In environmental science, it is commonly applied for measuring nitrates, phosphates, sulfates, and heavy metal complexes in water samples.
It is also useful in kinetic studies, where reaction progress is monitored by measuring absorbance changes over time.
Types of UV Visible Spectrophotometers
The instrumentation can be classified into two main types.
Single-Beam Spectrophotometer
In this type, light passes through one optical path. The blank and sample are measured separately. It is simple and cost-effective.
Double-Beam Spectrophotometer
This instrument splits light into two beams: one passes through the sample and the other through the reference. It provides better accuracy, improved baseline stability, and faster analysis.
Interpretation of UV Visible Spectrum
The output of UV-Visible spectroscopy is an absorption spectrum, which is a graph of absorbance versus wavelength.
The most important parameter is λmax, which represents the wavelength of maximum absorbance. It helps in:
- identifying compounds
- selecting analytical wavelengths
- studying structural features
- monitoring reaction changes
For quantitative analysis, a calibration curve is prepared using standards of known concentration. The absorbance of unknown samples is then compared with this curve to determine concentration accurately.
Common Solvents Used in UV Visible Spectroscopy
The choice of solvent is crucial because it must remain transparent in the desired wavelength range.
Commonly used solvents include:
- Water
- Methanol
- Ethanol
- Hexane
- Chloroform
Polar solvents are preferred for polar compounds, while non-polar solvents are used for hydrophobic analytes.
Advantages of UV Visible Spectroscopy
UV-Visible spectroscopy offers several important benefits. It is a rapid, sensitive, accurate, and non-destructive technique. The method requires only a small sample volume and minimal sample preparation. It is also highly versatile and suitable for a broad range of organic and inorganic compounds.
Because of its speed and simplicity, it is widely used in routine quality control and research laboratories.
Limitations of UV Visible Spectroscopy
Despite its usefulness, the technique has certain limitations.
It is mainly applicable to compounds containing chromophores, so molecules without UV-absorbing groups may not be detectable. Turbid or highly scattering samples can produce inaccurate results. The accuracy may also be affected by solvent impurities, pH changes, stray light, and instability of the light source. Additionally, deviations from Beer-Lambert law can occur at high concentrations.
Conclusion
UV-Visible spectroscopy remains a cornerstone analytical technique in modern science because of its precision, simplicity, and versatility. From drug analysis and biochemical quantification to environmental monitoring and materials characterization, its applications are extensive. The technique’s ability to provide both qualitative spectral fingerprints and accurate quantitative measurements makes it indispensable in academic research, industrial laboratories, and quality assurance systems.





