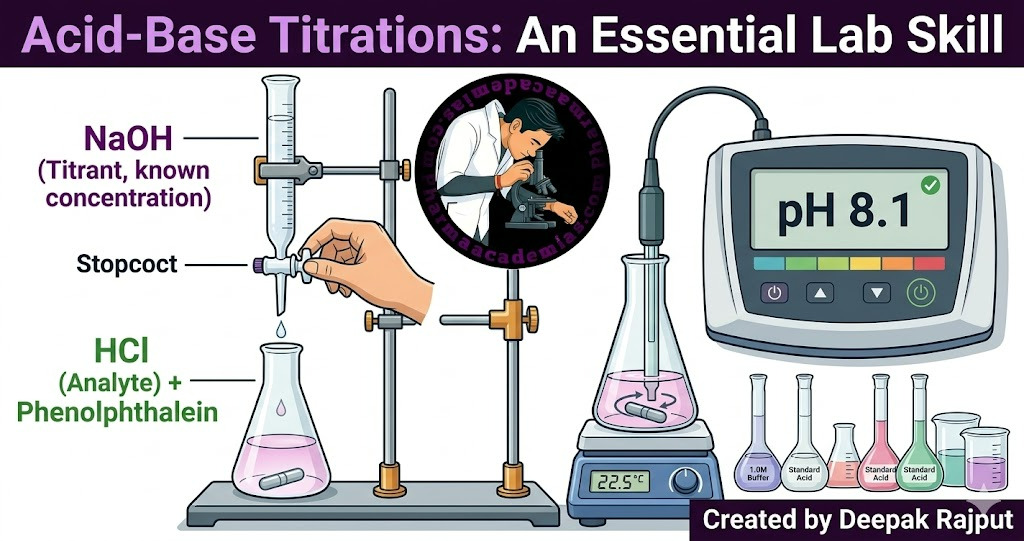
Acid-base titration is a quantitative analytical technique used to determine the unknown concentration of an acidic or basic solution by neutralizing it with a solution of known concentration. It is one of the most fundamental and widely used methods in analytical chemistry, pharmaceutical analysis, food chemistry, and environmental monitoring.
The process relies on the neutralization reaction between an acid and a base, producing a salt and water, which is a reaction with a stoichiometric relationship. The exact point at which the amount of acid is chemically equivalent to the amount of base is called the equivalence point. Detection of this point is crucial for accurate measurements, often achieved using color indicators or instrumental methods such as pH meters, conductometric probes, or potentiometric systems.
Mathematically, if (Va) is the volume of acid of concentration (Ca) and (Vb) is the volume of base of concentration (Cb), the relationship is expressed as:
na ⋅ Ca ⋅ Va = nb ⋅ Cb ⋅ Vb
Where (na) and (nb) are the stoichiometric coefficients of acid and base in the neutralization reaction.
History and Significance of Acid Base Titrations
The concept of titration was developed in the late 18th and early 19th centuries as chemists sought accurate methods for determining the concentration of chemical substances. French chemist Joseph Louis Gay-Lussac contributed to early volumetric methods, laying the foundation for modern acid-base titrations.
Acid-base titrations remain essential in laboratories worldwide for the analysis of pharmaceuticals, water quality, food products, and chemical formulations. They are also used in teaching laboratories to illustrate stoichiometry, equilibrium, and reaction kinetics.
Principle of Acid-Base Titration
The fundamental principle of acid-base titration is based on the neutralization reaction:
Acid + Base → Salt + Water
For example, the reaction between hydrochloric acid ((HCl)) and sodium hydroxide ((NaOH)) can be represented as:
HCl + NaOH → NaCl + H2O
At the equivalence point, the number of moles of (H^+) ions from the acid equals the number of moles of (OH^-) ions from the base. The endpoint—detected by a color change of an indicator or an instrumental reading—should closely match the equivalence point for accurate results.
Key Factors Affecting Titration:
- Strength of the acid and base (strong vs. weak)
- Choice of indicator
- Temperature and reaction medium
- Accuracy of volumetric glassware
Classification of Acid Base Titrations
Acid-base titrations can be classified based on reactant strength, reaction type, or method of endpoint detection.
1. Based on Strength of Reactants
a. Strong Acid vs. Strong Base
- Example: HCl titrated with NaOH
- Characteristics:
- Equivalence point at pH 7
- Sharp pH change near the equivalence point
- Methyl orange, phenolphthalein suitable indicators
b. Strong Acid vs. Weak Base
- Example: HCl titrated with NH₃ solution
- Characteristics:
- Equivalence point pH < 7
- Gradual rise in pH near endpoint
- Methyl orange commonly used
c. Weak Acid vs. Strong Base
- Example: Acetic acid (CH₃COOH) titrated with NaOH
- Characteristics:
- Equivalence point pH > 7
- Gradual pH rise near endpoint
- Phenolphthalein as preferred indicator
d. Weak Acid vs. Weak Base
- Example: Ammonium acetate titrated with acetic acid
- Characteristics:
- Equivalence point difficult to detect
- Gradual pH change; indicators less reliable
- pH meter or conductometric methods recommended
2. Based on Titration Methods
- Direct Titration: Standard solution is added directly to the analyte until neutralization. Most common method.
- Back Titration: An excess of titrant is added, then the remaining unreacted titrant is measured by titration. Useful for slow reactions or insoluble analytes.
- Conductometric Titration: Measures electrical conductivity change during the reaction, ideal when indicators fail.
- Potentiometric Titration: Uses a pH electrode or potentiometer to detect equivalence point electronically.
- Thermometric Titration: Measures the heat change during neutralization, applicable in industrial settings.
3. Based on Indicators Used
The choice of indicator depends on the type of titration:
- Methyl Orange: Strong acid vs. weak base (pH range 3.1–4.4)
- Phenolphthalein: Weak acid vs. strong base (pH range 8.3–10.0)
- Bromothymol Blue: Strong acid vs. strong base (pH range 6.0–7.6)
Applications of Acid Base Titrations
- Pharmaceutical Analysis: Determining the concentration of active ingredients in drugs such as aspirin, antacids, or vitamins.
- Water Quality Testing: Measuring alkalinity, acidity, and hardness in drinking water and industrial effluents.
- Food Industry: Analysis of citric acid, acetic acid, and ascorbic acid content in foods and beverages.
- Environmental Monitoring: Detecting pollutants and acid rain components in water and soil.
- Research and Education: Teaching stoichiometry, equilibrium, buffer solutions, and reaction kinetics.
Advantages of Acid-Base Titrations
- Highly accurate and reproducible results
- Simple and cost-effective
- Applicable to a wide variety of acids and bases
- Can be adapted for instrumental or automated titrations
- Provides a clear understanding of chemical stoichiometry
Tips for Accurate Titration
- Use clean and calibrated volumetric glassware (burette, pipette, and flask).
- Select an appropriate indicator based on the type of acid and base.
- Add titrant slowly near the endpoint to avoid overshooting.
- Perform multiple titrations for reproducibility and calculate the average volume.
- Maintain constant temperature to prevent variations in reaction rates.
Conclusion
Acid-base titration is a cornerstone of analytical chemistry, offering a precise and systematic method to quantify acids and bases. Its classification based on reactant strength, method, and indicator choice allows chemists to tailor the procedure for pharmaceutical, environmental, and industrial applications. Modern adaptations, such as potentiometric and conductometric titrations, have expanded the utility of this classical technique, making it a versatile tool in both educational and professional laboratories.
References
- Skoog, D.A., West, D.M., Holler, F.J., & Crouch, S.R. Fundamentals of Analytical Chemistry, 10th Edition, Cengage Learning, 2014.
- Harris, D.C. Quantitative Chemical Analysis, 10th Edition, W.H. Freeman, 2016.
- Vogel, A.I. Vogel’s Textbook of Quantitative Chemical Analysis, 5th Edition, Longman, 1996.
- Mendham, J., Denney, R.C., Barnes, J.D., Thomas, M.J.K. Vogel’s Quantitative Chemical Analysis, 7th Edition, Pearson, 2000.






2 Comments