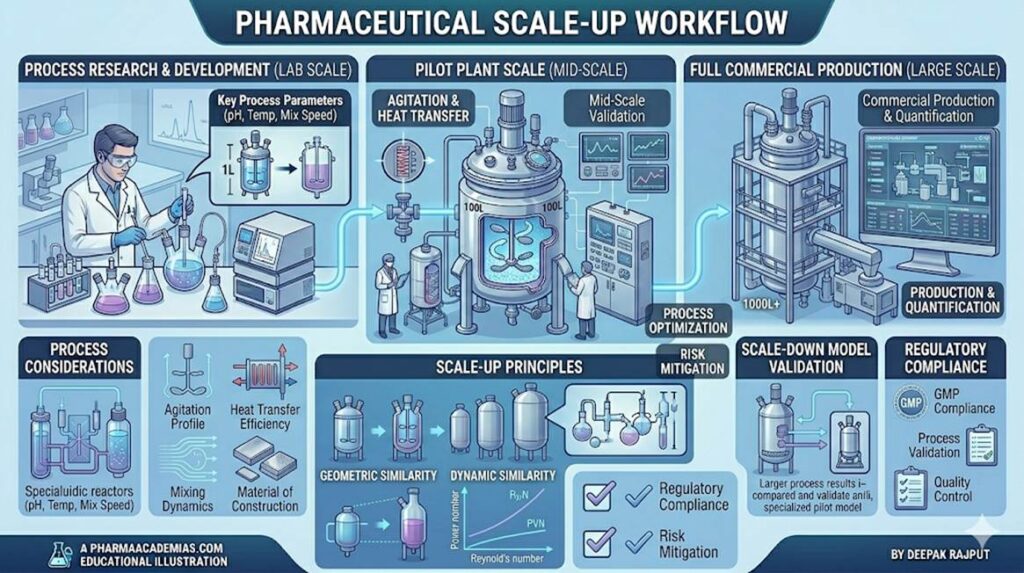
Pilot plant scale-up is a critical stage in pharmaceutical product development that bridges the gap between laboratory-scale formulation and full-scale industrial production. It involves the systematic enlargement of batch size and process conditions while maintaining product quality, safety, and efficacy.
The primary objective of pilot plant scale-up is to ensure that a formulation developed in the laboratory can be manufactured consistently on a larger scale without altering its critical quality attributes. This stage helps identify potential problems related to formulation, processing, equipment, and environmental factors before commercial production begins.
Scale-up is not simply increasing the batch size; rather, it requires careful optimization of process parameters, equipment selection, and operational conditions to ensure reproducibility and regulatory compliance.
General Considerations in Pilot Plant Scale Up Techniques
Successful scale-up requires a multidisciplinary approach involving formulation scientists, process engineers, quality assurance personnel, and production staff. Several critical factors must be considered to ensure smooth transition from laboratory to production scale.
Significance of Personnel Requirements of Pilot Plant Scale Up Techniques
Personnel play a vital role in the success of pilot plant operations. The complexity of pharmaceutical processes demands skilled and trained professionals capable of handling sophisticated equipment and maintaining strict quality standards.
Qualified personnel are required in areas such as formulation development, process engineering, quality control, quality assurance, and regulatory compliance. Each individual must have a clear understanding of Good Manufacturing Practices (GMP), standard operating procedures (SOPs), and safety protocols.
During scale-up, effective communication and coordination among different departments are essential. Any deviation in process or product quality must be quickly identified and addressed by trained staff.
In addition, continuous training programs are necessary to keep personnel updated with new technologies, regulatory guidelines, and operational procedures. Proper documentation practices must also be followed by personnel to ensure traceability and compliance with regulatory authorities.
Space Requirements in Pilot Plant Scale Up Techniques
Adequate space planning is essential for efficient pilot plant operations. The layout should be designed to ensure smooth workflow, minimize contamination risks, and comply with regulatory requirements.
The pilot plant should have sufficient space to accommodate equipment, raw materials, in-process materials, and finished products. It must allow proper movement of personnel and materials to avoid cross-contamination and ensure safety.
Different areas should be designated for specific operations such as dispensing, processing, packaging, quality control, and storage. These areas should be designed according to GMP guidelines with proper environmental controls such as temperature, humidity, and air filtration.
Utilities such as water supply, electricity, compressed air, and ventilation systems must be adequately installed and maintained. The design should also include provisions for waste disposal and sanitation.
Proper space allocation not only improves operational efficiency but also ensures compliance with regulatory standards and reduces the risk of errors during scale-up.
Also read this
QSEM and Q-Series Guidelines –
Raw Materials Considerations in Pilot Plant Scale Up Techniques
Raw materials are a crucial factor in pilot plant scale-up, as variations in their quality, source, or properties can significantly affect the final product.
All raw materials used in scale-up must meet predefined specifications and should be sourced from reliable and approved vendors. Changes in supplier or grade of material may lead to differences in physicochemical properties such as particle size, polymorphism, moisture content, and flow characteristics, which can affect processing and product performance.
It is important to evaluate the compatibility of raw materials with each other and with the manufacturing process. Stability studies should be conducted to ensure that materials do not degrade under processing conditions.
The scale-up process must also consider the availability of raw materials in large quantities for commercial production. Materials that are easily available at laboratory scale may not be feasible at industrial scale.
Proper storage conditions must be maintained to preserve the quality of raw materials. This includes control of temperature, humidity, and protection from light or contamination.
Additional General Considerations
Apart from personnel, space, and raw materials, several other factors influence pilot plant scale-up.
Process parameters such as mixing time, temperature, pressure, and equipment speed must be optimized and controlled. Equipment used in pilot scale should mimic industrial equipment as closely as possible to ensure reproducibility.
Validation of the process is essential to ensure consistency and reliability. Documentation, including batch manufacturing records and validation protocols, must be maintained according to regulatory requirements.
Quality control testing should be performed at every stage to ensure that the product meets specifications. Stability studies should also be conducted to predict shelf life and storage conditions.
Conclusion
Pilot plant scale-up is a crucial step in pharmaceutical manufacturing that ensures successful transition from laboratory formulation to commercial production. It requires careful consideration of personnel, space, and raw materials, along with process optimization and regulatory compliance.
Proper planning and execution of scale-up activities help in minimizing risks, improving product quality, and ensuring efficient large-scale manufacturing. A well-designed pilot plant not only enhances productivity but also plays a key role in achieving consistent and reliable pharmaceutical products.





