Colloidal dispersions are heterogeneous biphasic systems in which one substance is dispersed as extremely fine particles in another continuous medium. The dispersed particles are larger than those present in true solutions but smaller than those in coarse suspensions, generally lying in the range of 1 nm to 500 nm (or up to 1000 nm in some systems). Because of this intermediate particle size, colloidal dispersions show unique optical, kinetic, and electrical properties that make them highly important in physical pharmaceutics, drug delivery, suspensions, emulsions, gels, and nanoparticulate systems.
In pharmaceutical science, colloidal systems are especially important because they improve drug solubility, stability, absorption, targeted delivery, and controlled release behavior.
Classification of Colloidal Dispersions
The classification of colloidal dispersions can be explained in different ways depending on particle size, physical state, and interaction with the dispersion medium.
1) Classification Based on Particle Size in Dispersed Systems
Dispersed systems are broadly divided into molecular dispersions, colloidal dispersions, and coarse dispersions.
A molecular dispersion or true solution contains particles smaller than 1 nm, such as glucose or sodium chloride in water. These systems are completely homogeneous and do not scatter light.
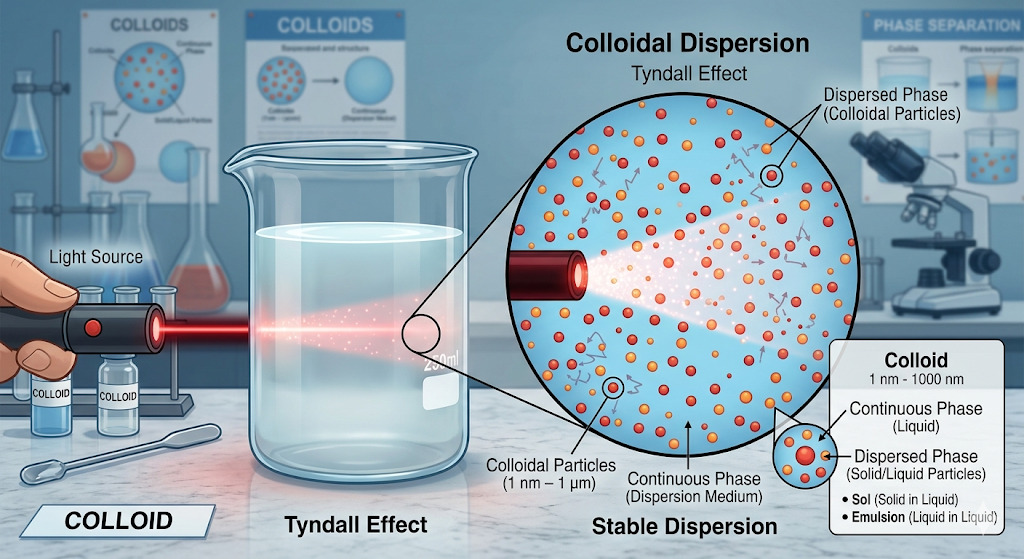
A colloidal dispersion contains particles generally in the range of 1–500 nm, which are too small to be seen by the naked eye but large enough to scatter light.
A coarse dispersion contains particles larger than 500 nm, such as suspensions and emulsions, where particles may settle on standing.
This classification shows that colloids occupy the intermediate region between true solutions and coarse dispersions.
2) Classification Based on Physical State of Dispersed Phase and Dispersion Medium
Depending on the physical states of the dispersed phase and the dispersion medium, colloidal systems may exist in several forms.
A sol is formed when a solid is dispersed in a liquid, such as gold sol or starch sol.
A gel is a semisolid system in which the dispersed phase forms a network throughout the liquid, such as gelatin gel.
An emulsion is a colloidal system where one liquid is dispersed in another immiscible liquid, such as milk.
A foam contains gas dispersed in liquid, such as shaving foam.
An aerosol contains solid or liquid particles dispersed in gas, such as inhalation sprays and smoke.
This type of classification is very useful in pharmaceutics because it directly relates to dosage forms.
3) Classification Based on Affinity for Dispersion Medium
Based on the interaction between dispersed particles and the medium, colloids are classified into lyophilic, lyophobic, and association colloids.
Lyophilic colloids are “solvent-loving” systems in which the dispersed phase has strong affinity for the medium. Examples include gelatin, acacia, and starch. These are usually stable and reversible.
Lyophobic colloids are “solvent-hating” systems with little affinity for the medium, such as metal sols and sulfur sols. These are comparatively less stable and may require stabilizers.
Association colloids or micellar colloids are formed by surfactants above a certain concentration called the critical micelle concentration (CMC), such as soap and detergent micelles. These are highly important in solubilization and drug delivery.
General Characteristics of Colloidal Dispersions
The general characteristics of colloidal dispersions arise mainly from their intermediate particle size and large surface area.
The first characteristic is particle size. Colloidal particles are small enough to remain dispersed for long periods but large enough to exhibit special interfacial properties.
The second important characteristic is the biphasic nature of the system. Every colloidal system contains:
- dispersed phase
- dispersion medium
This makes the system heterogeneous microscopically, although it may appear homogeneous macroscopically.
Another important characteristic is the large surface area. Since particles are very small, the total surface area exposed is extremely high. This increases:
- adsorption
- dissolution rate
- surface free energy
- reactivity
- catalytic activity
This property is highly beneficial in pharmaceutical formulations because it can improve drug solubility and bioavailability.
Surface charge: The next major characteristic is surface charge. Colloidal particles usually carry either a positive or negative charge due to ionization or adsorption of ions from the medium. This charge causes mutual repulsion between particles and prevents aggregation, thereby improving stability.
Tyndall effect: A highly characteristic feature is the Tyndall effect, which means colloidal particles scatter a beam of light passing through the system. This helps distinguish colloids from true solutions.
Brownian movement: Another important property is Brownian movement, the continuous zig-zag movement of particles due to bombardment by molecules of the dispersion medium. This motion helps prevent sedimentation and contributes to kinetic stability.
Colloidal dispersions also exhibit slow diffusion, high adsorption capacity, and low sedimentation tendency, making them much more stable than coarse suspensions.
They also possess kinetic stability but thermodynamic instability, meaning they may remain uniformly dispersed for long periods even though the system is not in the lowest energy state.
Pharmaceutical Importance of colloidal dispersions
In pharmacy, colloidal dispersions are widely used in:
- suspensions
- emulsions
- gels
- liposomes
- nanoparticles
- micelles
- parenteral delivery
- ophthalmics
- topical preparations
- targeted drug delivery
Their small size improves drug absorption, tissue penetration, controlled release, and organ targeting.
Conclusion
Colloidal dispersions are heterogeneous biphasic systems with particle size intermediate between true solutions and coarse dispersions.
They are classified on the basis of:
- particle size
- physical state
- affinity for medium
Their major general characteristics include small particle size, large surface area, surface charge, Tyndall effect, Brownian motion, slow diffusion, and high stability.
Because of these unique properties, colloidal dispersions hold a major place in physical pharmaceutics, advanced dosage forms, and modern drug delivery systems.