Buffers are solutions that resist changes in pH when small amounts of acid or alkali are added. They usually consist of a weak acid and its salt with a strong base, or a weak base and its salt with a strong acid. In pharmaceutical and biological systems, buffers are extremely important because the stability, solubility, compatibility, absorption, and safety of drugs as well as the normal functioning of body fluids depend on maintaining an optimum pH range.
In physical pharmaceutics and biopharmaceutics, buffers are among the most frequently discussed systems because even a small pH change can lead to drug degradation, precipitation, irritation, or reduced therapeutic action.
Buffers in Pharmaceutical Systems
In pharmaceutical preparations, buffers are used mainly to maintain the desired pH throughout the shelf life and during administration. Many drugs are stable only within a narrow pH range. If the pH shifts, the drug may undergo hydrolysis, oxidation, precipitation, color change, or loss of potency.
For this reason, buffer systems are incorporated into:
- ophthalmic solutions
- parenteral preparations
- oral liquids
- nasal drops
- topical solutions
- semisolid preparations
- biological products
- vaccine formulations
The most important role of pharmaceutical buffers is to maintain maximum drug stability. For example, certain antibiotics, vitamins, and protein drugs are highly sensitive to pH variation.
A second important role is improving solubility. Weakly acidic and weakly basic drugs often show pH-dependent solubility, so the buffer ensures that the drug remains dissolved and available for absorption.
Another major function is patient comfort and safety, especially in ophthalmic and injectable formulations. Solutions with unsuitable pH may cause pain, tissue irritation, lacrimation, corneal damage, or phlebitis. This is why eye drops are generally buffered close to tear pH, around 7.4.
Common pharmaceutical buffer systems include:
- Acetate buffer → acetic acid + sodium acetate
- Phosphate buffer → sodium dihydrogen phosphate + disodium hydrogen phosphate
- Citrate buffer → citric acid + sodium citrate
- Borate buffer → boric acid + sodium borate
Among these, phosphate buffer is one of the most widely used pharmaceutical and biological buffers because of its good buffering range near physiological pH.
Selection of Buffers in Pharmaceutical Formulations
The choice of a pharmaceutical buffer is highly scientific.
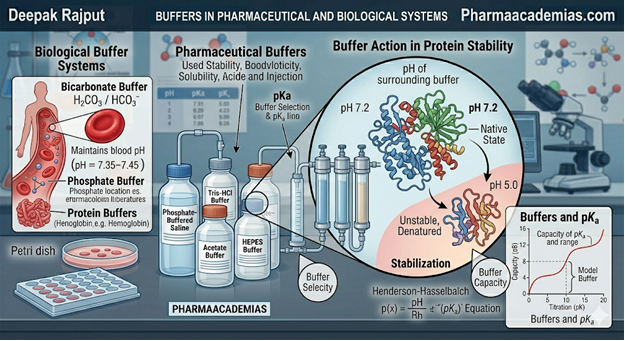
The buffer should have a pKa close to the desired formulation pH, because maximum buffering capacity occurs when pH = pKa
Other important factors considered include:
- compatibility with drug
- non-toxicity
- no interference with absorption
- adequate buffer capacity
- stability during storage
- physiological acceptability
- minimum tissue irritation
- compatibility with container and preservatives
A very high buffer capacity is usually avoided in ophthalmic and parenteral products because it may overpower the body’s own natural buffer systems.
Buffers in Biological Systems
In living organisms, buffers are essential for maintaining acid–base homeostasis. Even slight pH changes can alter:
- enzyme activity
- protein structure
- metabolic pathways
- oxygen transport
- nerve conduction
- muscle contraction
The human body therefore uses several highly efficient biological buffer systems.
1) Bicarbonate Buffer System
This is the most important extracellular buffer system, especially in blood plasma.
It consists of:
Carbonic acid (H₂CO₃) / Sodium bicarbonate (NaHCO₃)
This system maintains blood pH around 7.35–7.45 and is closely regulated by the lungs and kidneys.
It is the principal buffer system responsible for rapid compensation of acid–base imbalance.
2) Phosphate Buffer System
The phosphate system is highly important in:
- intracellular fluid
- renal tubular fluid
- urine
- cytoplasm
It consists of:
NaH₂PO₄ / Na₂HPO₄
This system is particularly effective around neutral pH and plays a major role in urinary acid excretion and intracellular enzyme reactions.
3) Protein Buffer System
Proteins contain amino and carboxyl groups that can accept or donate hydrogen ions.
Hence plasma proteins and intracellular proteins act as excellent buffers.
A major example is hemoglobin, which acts as a powerful buffer in red blood cells and helps maintain blood pH during transport of carbon dioxide and oxygen.
This system is extremely important in respiratory physiology and tissue metabolism.
Importance in Pharmacy and Medicine
The concept of buffers in pharmaceutical and biological systems is highly important because it directly influences:
- drug stability
- formulation elegance
- shelf life
- drug absorption
- patient comfort
- isotonicity adjustment
- blood pH maintenance
- renal acid excretion
- enzyme function
- metabolic balance
In modern dosage forms such as liposomes, monoclonal antibodies, vaccines, ophthalmics, and injectables, correct buffering is critical for therapeutic success.
Conclusion
Buffers in pharmaceutical and biological systems are chemical systems that resist pH change and help maintain the required hydrogen ion concentration for drug stability and physiological function.
In pharmaceuticals, they are used to improve stability, solubility, compatibility, and patient comfort, while in biological systems they maintain blood, intracellular, and urinary pH within normal limits.
The most important biological buffers are the bicarbonate, phosphate, and protein buffer systems, while common pharmaceutical buffers include acetate, citrate, phosphate, and borate buffers.
Because pH control is fundamental to both dosage form design and human physiology, buffers remain one of the most essential topics in physical pharmaceutics and biological sciences.