High Performance Liquid Chromatography (HPLC) is one of the most powerful and widely used analytical techniques for the separation, identification, and quantification of components in a mixture. It is an advanced form of liquid chromatography that uses high pressure to push a liquid mobile phase through a column packed with fine stationary phase particles, resulting in high resolution and efficient separation.
HPLC is extensively used in pharmaceutical analysis, clinical research, biotechnology, environmental analysis, and food industries. It is particularly important for analyzing non-volatile, thermally unstable, and polar compounds that cannot be analyzed by Gas Chromatography.
The technique has become a standard method in quality control laboratories due to its accuracy, reproducibility, and versatility.
Theory of High Performance Liquid Chromatography
The principle of HPLC is based on the distribution of analytes between two phases: a mobile phase (liquid solvent) and a stationary phase (solid adsorbent or bonded phase).
When a sample is injected into the system, it is carried by the mobile phase under high pressure through the column. The components of the sample interact differently with the stationary phase depending on their polarity, size, charge, and affinity.
As a result, each component travels at a different rate through the column. Compounds with lower affinity for the stationary phase elute faster, while those with stronger interactions elute later.
The separation mechanism may involve adsorption, partition, ion exchange, or size exclusion depending on the type of HPLC used.
Retention time (time taken by a compound to elute) is used for identification, while peak area is used for quantification.
Instrumentation of High Performance Liquid Chromatography
The HPLC system consists of several key components that work together to achieve efficient separation and detection.
1. Solvent Reservoir (Mobile Phase)
The solvent reservoir holds the mobile phase, which is usually a mixture of water and organic solvents such as methanol or acetonitrile. The composition of the mobile phase is carefully selected to optimize separation.
2. Degasser
The degasser removes dissolved gases (oxygen, nitrogen, carbon dioxide) from the mobile phase. This is important because dissolved gases can form bubbles that interfere with detector response and flow stability.
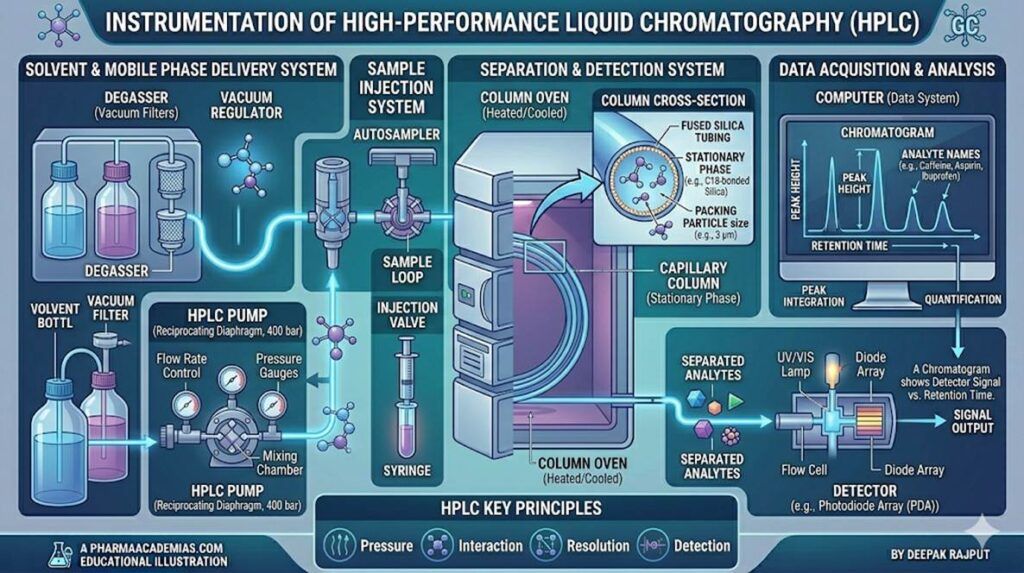
3. Pump
The pump is one of the most critical components of HPLC. It delivers the mobile phase at high pressure (typically 100–400 bar or more) with a constant and precise flow rate. This high pressure is necessary because the column is tightly packed with fine particles that create resistance to flow.
There are different types of pumps such as reciprocating pumps and syringe pumps, with reciprocating pumps being most commonly used.
4. Injector System
The injector introduces a precise volume of sample into the flowing mobile phase without interrupting the system pressure. A sample loop system is commonly used for high reproducibility.
5. Column
The column is the heart of the HPLC system where separation occurs. It is typically made of stainless steel and packed with small particles (3–10 µm).
Common stationary phases include silica-based materials bonded with different functional groups such as C18 (reverse phase), C8, or polar phases.
Columns can be classified as:
- Normal phase columns (polar stationary phase)
- Reverse phase columns (non-polar stationary phase)
- Ion exchange columns
- Size exclusion columns
Reverse phase HPLC is the most widely used mode.
6. Detector
The detector identifies and quantifies separated components as they elute from the column. It converts chemical signals into electrical signals to generate a chromatogram.
Common detectors include:
- UV-Visible detector (most widely used)
- Photodiode array (PDA) detector
- Fluorescence detector
- Refractive index detector
- Mass spectrometry (LC-MS)
The UV detector works on the principle of light absorption by analytes at specific wavelengths.
7. Data System (Recorder)
The data system processes detector signals and generates a chromatogram showing peaks corresponding to different compounds. It is also used for integration, quantification, and reporting.
Advantages of HPLC
HPLC offers several important advantages that make it a preferred analytical technique.
It provides high resolution and excellent separation efficiency even for complex mixtures. It can analyze a wide range of compounds including non-volatile, polar, thermally unstable, and high molecular weight substances.
HPLC offers high sensitivity and accurate quantification with excellent reproducibility. It requires relatively small sample volumes and provides rapid analysis compared to traditional liquid chromatography methods.
The technique is highly versatile, as different stationary phases and mobile phase compositions can be used to optimize separation. It is also suitable for both qualitative and quantitative analysis.
Coupling with mass spectrometry (LC-MS) further enhances its capability for structural identification of unknown compounds.
Applications of HPLC
HPLC has wide-ranging applications across pharmaceutical, clinical, environmental, and industrial fields.
- In pharmaceutical analysis, HPLC is used for drug purity testing, assay of active pharmaceutical ingredients (APIs), stability studies, dissolution testing, and impurity profiling.
- In clinical and biomedical research, it is used for analysis of drugs in biological fluids such as blood and plasma, as well as biomarker detection and therapeutic drug monitoring.
- In environmental analysis, HPLC helps detect pesticides, herbicides, and pollutants in water and soil samples.
- In the food industry, it is used for analysis of preservatives, vitamins, additives, and contaminants.
- In biotechnology, HPLC is used for protein purification, peptide mapping, and nucleic acid analysis.
It is also widely used in forensic science for drug abuse testing and toxicology studies.
Conclusion
High Performance Liquid Chromatography (HPLC) is a highly efficient, accurate, and versatile analytical technique used for the separation and analysis of a wide range of compounds. Its ability to analyze non-volatile and thermally unstable substances makes it superior to many other chromatographic techniques.
Due to its high sensitivity, reproducibility, and broad applications in pharmaceuticals, clinical diagnostics, and environmental science, HPLC has become an indispensable tool in modern analytical laboratories.