Volatile oils, also known as essential oils, are complex mixtures of aromatic compounds obtained from plants, possessing distinctive odors and therapeutic properties. These oils are highly hydrophobic, volatile, and sensitive to heat, which makes their isolation a critical step in pharmaceutical and perfumery industries. They are widely used in aromatherapy, pharmaceutical formulations, flavorings, and as antimicrobial or anti-inflammatory agents.
The isolation of volatile oils is a delicate process, as improper techniques can lead to loss of active constituents, degradation, or change in fragrance and efficacy. The aim of isolation is to extract these oils efficiently while preserving their chemical composition and bioactivity.
Definition of Isolation of Volatile Oils
The isolation of volatile oils refers to the process of separating the essential oil from the plant material while retaining its aromatic, chemical, and therapeutic properties. This process can be achieved through physical, mechanical, or chemical methods, depending on the nature of the plant and the desired purity of the oil.
Principles of Isolation
Volatile oils are primarily composed of terpenes, alcohols, aldehydes, ketones, phenols, and esters, which are sensitive to heat, light, and air. The principle behind their isolation involves separating these lipophilic, volatile components from non-volatile plant matter without altering their chemical structure.
The methods of isolation rely on:
- Volatility: Volatile oils have low boiling points, allowing distillation techniques to separate them from water or steam.
- Solubility: Their lipophilic nature allows extraction using organic solvents that selectively dissolve the oil components.
- Mechanical release: Certain plant materials, like citrus peels, allow mechanical pressing or expression to release oil.
Methods of Isolation of Volatile Oils
The choice of method depends on the plant source, oil composition, and intended use. The main methods are:
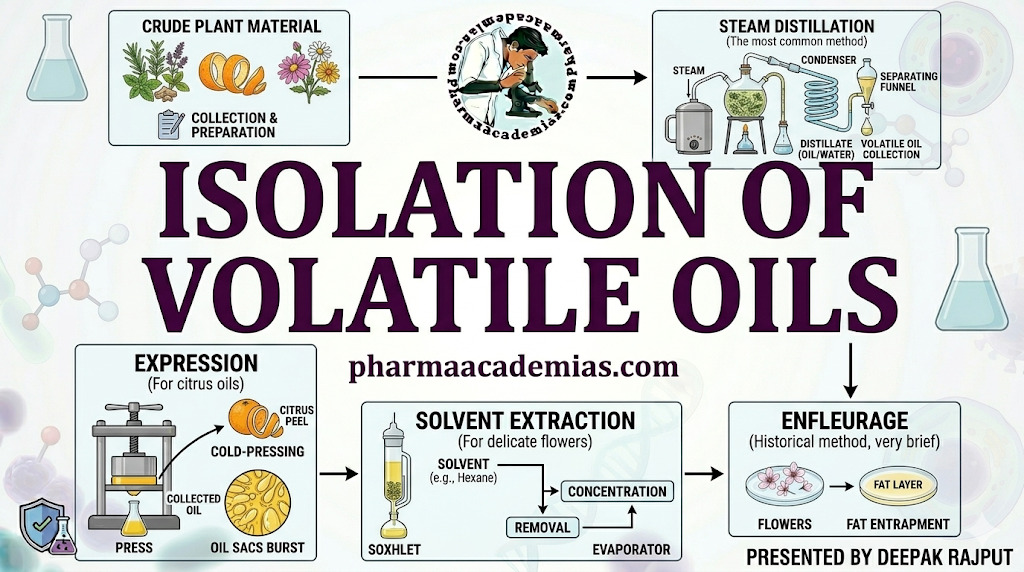
- Steam Distillation
Steam distillation is the most common method for isolating volatile oils from leaves, flowers, and roots. The plant material is subjected to steam, which volatilizes the oil, allowing it to be condensed and separated from water in a Clevenger-type apparatus. This method is particularly suitable for heat-stable oils and preserves the fragrance and therapeutic properties.
- Hydrodistillation
In hydrodistillation, the plant material is immersed in water and boiled, with the volatile oil co-distilling with water vapor. The condensed mixture is then decanted or separated using a separating funnel. Hydrodistillation is useful for flowers and delicate plant parts, where direct steam could damage the oil.
- Expression (Cold Pressing)
Expression is a mechanical method mainly used for citrus peels, such as orange, lemon, or bergamot. The oil is released by pressing or grating the plant material, followed by separation from juice and water. This method avoids heat, making it ideal for thermally sensitive oils.
- Solvent Extraction
Solvent extraction is employed when steam distillation is inefficient or the oil is highly sensitive to heat. Non-polar solvents, such as hexane, petroleum ether, or ethanol, dissolve the essential oils, which are then separated by evaporation of the solvent. This method is used for flowers and delicate plant tissues, like jasmine or rose.
- Enfleurage
Enfleurage is an old, traditional method where odoriferous plant material is placed on odorless fats, allowing the oil to diffuse into the fat. The fat is later washed with alcohol to extract the oil, leaving behind the non-volatile residue. This method is rarely used today due to high cost and low efficiency, but it is ideal for extremely delicate flower oils.
- Supercritical Fluid Extraction
This modern technique uses supercritical CO₂ as a solvent to extract volatile oils. The method is highly efficient, selective, and preserves heat-sensitive constituents. It is increasingly used in the pharmaceutical, cosmetic, and food industries for high-purity essential oils.
Factors Affecting Isolation of Volatile Oils
The efficiency of volatile oil isolation is influenced by multiple factors, including:
- Plant material quality: Fresh, healthy, and properly dried plant materials yield higher oil content.
- Moisture content: Excessive moisture may dilute the oil or cause hydrolysis.
- Temperature: High temperatures may degrade heat-sensitive components.
- Time of collection: Oils often vary seasonally or diurnally, affecting both quantity and fragrance.
- Method of isolation: Each technique preserves different chemical constituents to varying degrees.
Pharmaceutical Applications of Volatile Oils
Volatile oils have extensive applications in pharmacy:
- They act as therapeutic agents, including antimicrobial, anti-inflammatory, carminative, and spasmolytic activities.
- They are used in aromatic medicines and phytopharmaceutical formulations.
- Volatile oils serve as flavoring and fragrance agents in syrups, lozenges, and ointments.
- They are employed in controlled-release formulations, where their hydrophobic nature allows encapsulation in lipid-based carriers.
- Essential oils are also incorporated in topical preparations for antiseptic, analgesic, or aromatic purposes.
Advantages of Proper Isolation
Effective isolation techniques ensure that:
- The therapeutic and aromatic properties of the oil are preserved.
- Contaminants and non-volatile components are removed, enhancing purity.
- The oil is stable for storage, formulation, and industrial use.
- Accurate dosing in pharmaceutical preparations is achievable, ensuring therapeutic efficacy.
Conclusion
Isolation of volatile oils is a critical process in pharmaceutical sciences, as it allows for extraction of potent bioactive compounds from plant sources while maintaining chemical integrity, fragrance, and therapeutic efficacy. Mastery of the different isolation methods, their principles, and applications is essential for pharmacy students, researchers, and formulation scientists, particularly in the development of herbal medicines, aromatic formulations, and essential oil-based therapeutics.
Key Points for Study
- Volatile oils are hydrophobic, aromatic, and thermally sensitive.
- Common isolation methods: steam distillation, hydrodistillation, expression, solvent extraction, enfleurage, supercritical fluid extraction.
- Factors affecting yield include plant quality, moisture, temperature, time of collection, and extraction method.
- Pharmaceutical applications: therapeutics, aromatherapy, flavoring, antiseptics, and topical formulations.
References
- Trease, G.E., Evans, W.C. Pharmacognosy, 16th Edition, Elsevier, 2009.
- Kokate, C.K., Purohit, A.P., Gokhale, S.B. Pharmacognosy, 54th Edition, Nirali Prakashan, 2021.
- Harborne, J.B. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis, 3rd Edition, Springer, 1998.
- Rowe, R.C., Sheskey, P.J., Quinn, M.E. Handbook of Pharmaceutical Excipients, 6th Edition, 2009.






