Volatile oils, commonly referred to as essential oils, are naturally occurring, aromatic, and highly volatile substances obtained from various plant parts such as leaves, flowers, bark, seeds, and roots. These oils are complex mixtures primarily composed of terpenoids (monoterpenes and sesquiterpenes) along with phenylpropanoids and oxygenated derivatives like alcohols, aldehydes, ketones, esters, and ethers.
Their importance in pharmaceutical sciences, aromatherapy, perfumery, and food industries makes their correct identification essential. Since volatile oils are prone to oxidation, degradation, and adulteration, a robust identification system is necessary to ensure quality, purity, safety, and therapeutic efficacy.
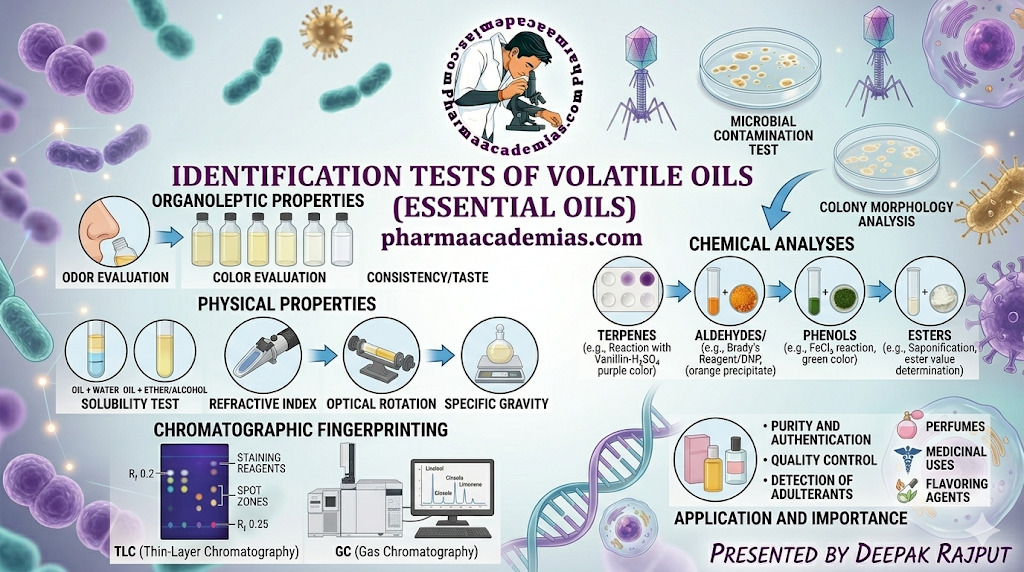
From an pharmacy perspective, the identification of volatile oils is not limited to simple observation but extends to advanced analytical techniques such as chromatography and spectroscopy, supported by classical chemical tests.
Organoleptic Identification Tests of Volatile Oils
The primary and most immediate method of identifying volatile oils is through organoleptic evaluation, which involves sensory perception. Each volatile oil possesses a unique aroma profile, which arises from its specific chemical composition. For instance, clove oil exhibits a strong, spicy, and pungent odor due to the presence of eugenol, while peppermint oil imparts a cooling and refreshing sensation because of menthol.
The color and appearance of volatile oils also provide valuable clues. Freshly distilled oils are generally colorless or pale yellow, but prolonged storage or exposure to light and air can lead to darkening due to oxidation or polymerization reactions. Although taste can sometimes be used as an identification parameter, it is rarely employed in modern pharmaceutical analysis due to safety concerns.
Organoleptic evaluation, while simple, requires trained expertise and is typically used as a preliminary step before more sophisticated analytical methods.
Physical Identification Tests of Volatile Oils
Physical constants play a crucial role in the identification and standardization of volatile oils. These parameters are highly sensitive to compositional changes and therefore serve as reliable indicators of purity.
Specific gravity: The specific gravity of a volatile oil is determined using instruments such as a pycnometer or digital densitometer. Each oil has a characteristic density range; for example, clove oil is heavier than water, whereas most oils like turpentine are lighter. Any deviation from standard values suggests possible adulteration.
Refractive index: The refractive index, measured using an Abbe refractometer, reflects how light bends when passing through the oil. This property depends on the molecular structure and concentration of constituents. Even minor changes in composition can significantly alter the refractive index, making it a sensitive parameter for quality control.
Optical rotation: Another important property is optical rotation, which is measured using a polarimeter. Many volatile oil constituents are optically active due to the presence of chiral centers. The direction and magnitude of rotation provide insight into the stereochemical nature of the oil and help detect synthetic adulterants or racemization.
Solubility behavior further aids in identification. Volatile oils are generally insoluble in water but readily soluble in organic solvents and alcohol. The clarity or turbidity of solutions in alcohol can indicate the presence of impurities, resins, or fixed oils.
Chemical Identification Tests for Volatile Oils
Chemical tests are designed to detect specific functional groups present in volatile oils and provide qualitative confirmation of their constituents.
Phenolic compounds: The presence of phenolic compounds, such as eugenol in clove oil, can be confirmed by the ferric chloride test, which produces a characteristic blue or green coloration. Similarly, aldehydes like cinnamaldehyde in cinnamon oil respond to Schiff’s reagent, yielding a pink or magenta color, indicating the presence of a reactive carbonyl group.
Ketonic compounds: Ketonic compounds, such as carvone found in caraway oil, can be identified using 2,4-dinitrophenylhydrazine, which forms a yellow or orange precipitate due to hydrazone formation. Alcoholic constituents react with Lucas reagent, producing turbidity that varies depending on the class of alcohol.
Esters: Esters, which are common in many fragrant oils like lavender oil, are detected using the hydroxamic acid test, where a colored complex forms in the presence of ferric ions. Unsaturation, a key feature of many terpenes, is demonstrated by the decolorization of bromine water, indicating the presence of double bonds.
Salkowski reaction: The Salkowski reaction is particularly useful for detecting terpenes, producing a reddish-brown coloration upon treatment with concentrated sulfuric acid. These classical chemical tests remain valuable for preliminary identification and teaching laboratories, although they are increasingly complemented by instrumental techniques.
Chromatographic Identification of Volatile Oils
Chromatography represents one of the most powerful tools for the identification and analysis of volatile oils. Thin Layer Chromatography (TLC) is widely used for generating a chemical fingerprint profile. In this technique, the oil is applied to a silica gel plate and developed in an appropriate solvent system. The separated components appear as distinct spots with characteristic Rf values, which can be visualized under ultraviolet light or after spraying with detecting reagents.
Gas Chromatography (GC), however, is considered the gold standard for volatile oil analysis. It separates individual constituents based on their volatility and interaction with the stationary phase. When coupled with mass spectrometry (GC-MS), it provides detailed information about the molecular structure and identity of each component, allowing precise detection of even minor constituents.
GC-MS is particularly important in detecting adulteration, as it can reveal the presence of synthetic compounds or substitution with cheaper oils. The technique generates a unique chromatographic pattern that acts as a chemical signature for each essential oil.
Spectroscopic Methods for Identification
Spectroscopic techniques offer advanced and highly reliable methods for identifying volatile oils at a molecular level. Infrared (IR) spectroscopy is commonly used to detect functional groups by analyzing characteristic absorption bands. For example, hydroxyl groups show broad peaks, while carbonyl groups exhibit sharp absorptions.
Ultraviolet (UV) spectroscopy is less commonly used but can provide information about conjugated systems within the oil. Nuclear Magnetic Resonance (NMR) spectroscopy is one of the most sophisticated techniques, offering detailed insights into molecular structure, including the arrangement of atoms and functional groups.
These methods are indispensable in modern research and industrial quality control, where accuracy and reproducibility are critical.
Chemical Constants and Their Significance
Chemical constants provide quantitative measures that help in the evaluation of volatile oil quality. The acid value indicates the presence of free acids and increases with degradation. The ester value reflects the amount of esterified components, which are often responsible for the pleasant aroma of oils.
The saponification value gives an estimate of total ester content, while the iodine value indicates the degree of unsaturation. These constants are essential in detecting aging, oxidation, and adulteration, and are frequently included in pharmacopeial standards.
Pharmacopoeial and Special Tests
Certain volatile oils have specific identification tests described in pharmacopeias. For example, clove oil reacts with potassium hydroxide to form crystalline potassium eugenolate, confirming the presence of eugenol. Peppermint oil exhibits a characteristic cooling sensation and may crystallize upon cooling due to menthol. Eucalyptus oil can form crystalline derivatives with phosphoric acid, aiding in its identification.
Such tests are particularly useful in routine analysis and serve as confirmatory procedures.
Detection of Adulteration in Volatile Oils
Adulteration is a major concern in the essential oil industry. Common adulterants include fixed oils, synthetic chemicals, and cheaper volatile oils. Detection involves a combination of physical, chemical, and instrumental methods.
Changes in specific gravity, refractive index, or optical rotation often indicate adulteration. Chromatographic techniques like GC-MS provide definitive evidence by identifying unexpected components. TLC fingerprinting also helps in detecting deviations from standard profiles.
Ensuring authenticity is crucial not only for therapeutic efficacy but also for regulatory compliance and consumer safety.
Conclusion
The identification of volatile oils is a comprehensive process that integrates classical methods with modern analytical techniques. While organoleptic and physical tests provide initial insights, chemical tests confirm the presence of functional groups, and chromatographic and spectroscopic methods offer precise and reliable identification.
For pharmacy students and professionals in pharmacognosy and pharmaceutical analysis, mastering these techniques is essential for ensuring the quality, safety, and standardization of herbal products. With the advancement of analytical technologies, the identification of volatile oils has become more accurate and sophisticated, yet the fundamental principles remain rooted in classical pharmaceutical sciences.






