The D/L system of nomenclature is a method used to describe the configuration of optical isomers, particularly for carbohydrates and amino acids. It was first introduced by Emil Fischer in the 19th century and is based on the comparison of optical isomers to a reference compound, glyceraldehyde. The system does not indicate the direction of optical rotation (i.e., whether the compound is dextrorotatory or levorotatory); rather, it denotes the spatial configuration of the molecule.
Historical Basis: Glyceraldehyde
The D/L system was originally based on the orientation of the functional groups around the chiral center of glyceraldehyde, a simple sugar with the molecular formula (C3H6O3). Glyceraldehyde has one chiral carbon atom and exists as two enantiomers:
D-(+)-Glyceraldehyde: The hydroxyl group (-OH) on the right side of the chiral carbon in Fischer projection.
L-(-)-Glyceraldehyde: The hydroxyl group (-OH) on the left side of the chiral carbon in Fischer projection.
2. Understanding Fischer Projections
Fischer projections are a two-dimensional representation of a molecule, where:
The vertical lines represent bonds going away from the viewer (behind the plane of the paper).
The horizontal lines represent bonds coming towards the viewer (out of the plane of the paper).
In Fischer projections:
For carbohydrates, the reference point is the penultimate carbon (the second-to-last carbon atom).
For amino acids, the reference point is the α-carbon (the carbon to which the amino group, carboxyl group, and side chain are attached).
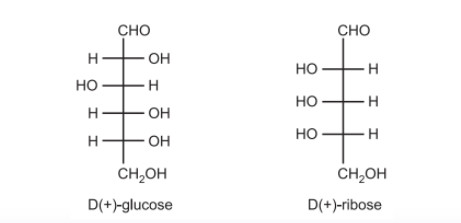
In the case of carbohydrates, particularly sugars like glucose and fructose, the D/L system is assigned based on the orientation of the hydroxyl group (-OH) on the penultimate carbon (the carbon farthest from the carbonyl group but closest to the -CH₂OH group):
D-isomer: The hydroxyl group (-OH) on the penultimate carbon is on the right side in the Fischer projection.
L-isomer: The hydroxyl group (-OH) on the penultimate carbon is on the left side in the Fischer projection.
Example: D-Glucose vs. L-Glucose:
D-Glucose: The hydroxyl group on the penultimate carbon (C5) is on the right.
L-Glucose: The hydroxyl group on the penultimate carbon (C5) is on the left.
Dand L-Nomenclature for Amino Acids
For amino acids, the D/L designation is based on the arrangement around the α-carbon. The orientation of the amino group (-NH₂) relative to the Fischer projection determines whether the amino acid is D or L.
D-amino acid: The amino group (-NH₂) is on the right side of the Fischer projection.
L-amino acid: The amino group (-NH₂) is on the left side of the Fischer projection.
Example: L-Alanine vs. D-Alanine:
L-Alanine: The amino group is on the left in the Fischer projection.
D-Alanine: The amino group is on the right in the Fischer projection.
Relation to Optical Activity
It is important to note that the Dand Lprefixes do not correspond directly to the optical activity of the compound (whether it rotates plane-polarized light to the right or left). For example:
D-(+)-glyceraldehyde rotates light to the right, hence the (+) sign, but other D-compounds may rotate light to the left.
L-(-)-glyceraldehyde rotates light to the left, but other L-compounds may rotate light to the right.
Therefore, the D/L system only indicates the relative configuration of a molecule compared to glyceraldehyde, not its optical rotation.
Applications of D/L Nomenclature
Carbohydrates: Most naturally occurring sugars (e.g., glucose, fructose) are in the D-form.
Amino Acids: Most naturally occurring amino acids (except some found in bacterial cell walls and antibiotics) are in the L-form.
The D/L system is widely used in biochemistry, particularly for sugars and amino acids, but it has been largely replaced by the R/S system of nomenclature for other organic compounds because the R/S system provides a more universal way of designating the absolute configuration of chiral centers.
Comparison with R/S Nomenclature
The R/S system (Cahn-Ingold-Prelog rules) is used for determining absolute configuration based on priority rules and is more versatile for complex molecules with multiple stereocenters.
The D/L system is a relative system based on the structure of glyceraldehyde and is mainly used for carbohydrates and amino acids.
Summary of D/L Nomenclature
D-isomer: Hydroxyl group (for sugars) or amino group (for amino acids) on the right in Fischer projection.
L-isomer: Hydroxyl group (for sugars) or amino group (for amino acids) on the left in Fischer projection.
Despite its limitations, the D/L system remains important in the study of biochemistry and molecular biology due to its historical significance and its practical application in describing the chirality of biologically relevant molecules.





