Proof Spirit: Pharmaceutical calculations form the backbone of safe drug formulation and dispensing. Among these, proof spirit, isotonic solutions, and dilute solutions (expressed as percentage and ratio strength) are fundamental concepts. They are widely applied in pharmaceutics, hospital pharmacy, and industrial formulation, and are frequently asked in GPAT, university exams, and viva.

Proof Spirit
Proof spirit is a traditional system used to express the strength of ethyl alcohol (ethanol). In the British system, which is followed in India, proof spirit is defined as a solution containing 57.1% v/v alcohol, and this is considered 100° proof.
The concept originated from early methods of testing alcohol strength, but it remains relevant in pharmaceutical calculations, especially for alcohol-based preparations.
Mathematical Relationship
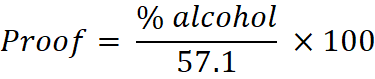
This formula allows easy conversion between percentage strength and proof value.
Interpretation of Proof Values
- When proof is greater than 100°, the solution is over proof (O.P.)
- When proof is less than 100°, it is under proof (U.P.)
For example, 80% alcohol corresponds to about 140° proof, meaning it is 40° over proof.
Practical Significance
Proof spirit is important in pharmacy because:
- It is used in preparation of tinctures, spirits, and extracts
- It helps in alcohol dilution calculations
- It appears in pharmacopoeial standards (IP/BP)
In practical compounding, pharmacists often need to adjust alcohol strength, and proof calculations make this process systematic and accurate.
Isotonic Solutions
An isotonic solution is one that exerts the same osmotic pressure as body fluids, such as blood plasma, tears, or intracellular fluid. Because of this balance, there is no net movement of water across cell membranes, and cells remain stable.
Biological Importance
The concept of isotonicity is crucial when solutions come into direct contact with body tissues. If the solution is not isotonic, it can cause cellular damage:
- Hypotonic solutions → water enters cells → swelling and hemolysis
- Hypertonic solutions → water leaves cells → shrinkage (crenation)
Thus, isotonicity is essential for maintaining cell integrity and physiological compatibility.
Common Isotonic Solutions
- 0.9% w/v Sodium Chloride solution (normal saline)
- 5% w/v Dextrose solution
These solutions are widely used because they closely match the osmotic pressure of blood plasma.
Methods of Adjusting Isotonicity
Several calculation methods are used in pharmaceutics:
- Freezing point depression method:
Based on the fact that body fluids freeze at approximately −0.52°C - Sodium chloride equivalent (E-value) method:
Expresses how much NaCl is equivalent to a given drug - White–Vincent method:
Used to calculate the volume of isotonic solution required
These methods are particularly important in preparing injectables and ophthalmic solutions, where even small deviations can cause irritation or damage.
Applications
Isotonic solutions are essential in:
- Intravenous (IV) fluids
- Eye drops and ophthalmic preparations
- Nasal and parenteral formulations
Dilute Solutions (Percentage and Ratio Strength)
Dilute solutions contain relatively low concentrations of solute, and in pharmacy, their strength is expressed either as percentage strength or ratio strength.
Percentage Strength
Percentage strength is the most common method of expressing concentration. It can be represented in three forms depending on the nature of solute and solvent:
- % w/v (weight in volume): grams of solute per 100 mL solution
- % v/v (volume in volume): mL of solute per 100 mL solution
- % w/w (weight in weight): grams of solute per 100 g solution
The general formula is:
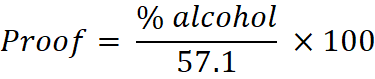
For example, a 5% w/v solution contains 5 g of drug in 100 mL of solution.
Ratio Strength
Ratio strength expresses concentration in the form 1 in x, meaning one part of solute is present in x parts of solution. This method is especially useful for very dilute preparations.
Examples include:
- 1:100 → 1 g in 100 mL
- 1:1000 → 1 g in 1000 mL
Relationship Between Percentage and Ratio
There is a simple relationship between the two systems:
- 1% = 1:100
- 0.1% = 1:1000
- 10% = 1:10
Thus, a 2% solution can be expressed as 1:50, which is often easier to interpret in practice.
Pharmaceutical Importance
Dilute solutions are widely used in:
- Antiseptic and disinfectant preparations
- Syrups, elixirs, and suspensions
- Topical formulations and lotions
Accurate expression of concentration ensures therapeutic efficacy and patient safety, especially when dealing with potent drugs.
Conclusion
Proof spirit, isotonic solutions, and dilute solution expressions are essential pillars of pharmaceutical calculations. Proof spirit standardizes alcohol strength, isotonic solutions ensure compatibility with body fluids, and percentage and ratio expressions provide flexible ways to represent concentration. A strong understanding of these concepts enables pharmacists to perform accurate calculations, prepare safe formulations, and maintain high standards of patient care.






