Cis–trans isomerism is a type of geometrical isomerism that occurs when there is restricted rotation around a double bond (C=C) or within cyclic compounds. In these molecules, atoms or groups attached to the carbon atoms cannot rotate freely, leading to different spatial arrangements.
Although cis and trans isomers have the same molecular formula and connectivity of atoms, they differ in the arrangement of substituent groups in space, which results in differences in physical and sometimes chemical properties.
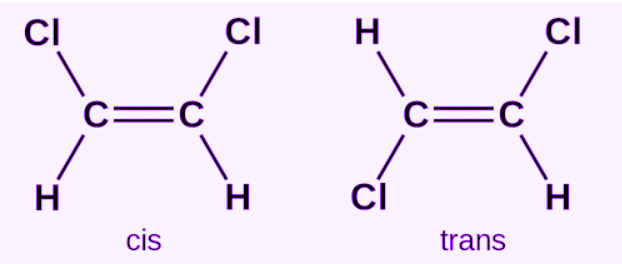
Conditions Required for Cis–Trans Isomerism
For cis–trans isomerism to occur, two main conditions must be satisfied:
1. Presence of Restricted Rotation
There must be restricted rotation, usually due to:
- Carbon–carbon double bond (C=C)
- Ring structures (cycloalkanes)
In a double bond, the π bond prevents free rotation, making the spatial arrangement fixed.
2. Different Groups on Each Carbon
Each carbon atom of the double bond must have two different substituents.
Example:
CH₃–CH = CH–CH₃
(But-2-ene)
Each carbon is attached to:
- H
- CH₃
Thus, cis–trans isomerism is possible.
Definition of Cis and Trans
1. Cis Isomer
When similar groups or higher priority groups are on the same side of the double bond, the compound is called the cis isomer.
Example: cis-but-2-ene
Structure (simplified):
CH₃ CH₃
\ /
C = C
/ \
H H
Both CH₃ groups are on the same side of the double bond.
2. Trans Isomer
When similar groups are on opposite sides of the double bond, the compound is called the trans isomer.
Example: trans-but-2-ene
Structure:
CH₃ H
\ /
C = C
/ \
H CH₃
The CH₃ groups are opposite each other.
Example of Cis–Trans Isomerism
Example: But-2-ene
Molecular Formula: C₄H₈
Two geometrical isomers exist:
- Cis-but-2-ene
- Trans-but-2-ene
Both compounds have:
- Same molecular formula
- Same structure
But they differ in spatial arrangement of groups around the double bond.
Cis–Trans Isomerism in Cyclic Compounds
Cis–trans isomerism also occurs in cyclic compounds because rotation around the ring is restricted.
Example: 1,2-dimethylcyclohexane
Cis Isomer
Both CH₃ groups are on the same side of the ring.
Trans Isomer
The CH₃ groups are on opposite sides of the ring.
Differences Between Cis and Trans Isomers
| Property | Cis Isomer | Trans Isomer |
| Arrangement | Same side | Opposite sides |
| Polarity | Usually polar | Usually non-polar |
| Boiling point | Higher | Lower |
| Melting point | Lower | Higher |
| Stability | Less stable | More stable |
Reason
In cis isomers, similar groups are closer together, causing steric repulsion, which decreases stability.
In trans isomers, groups are farther apart, making the molecule more stable.
Importance of Cis–Trans Isomerism
1. Pharmaceutical Importance
Different geometrical isomers may have different biological activities.
Example:
- Cisplatin – effective anticancer drug
- Transplatin – much less effective
2. Difference in Physical Properties
Cis and trans isomers differ in:
- Melting point
- Boiling point
- Solubility
- Dipole moment
3. Biological Selectivity
Biological molecules such as enzymes and receptors are stereospecific, meaning only one isomer may show the desired biological effect.
Summary
Cis–trans isomerism is a type of geometrical isomerism that arises due to restricted rotation around double bonds or in cyclic structures. In cis isomers, similar groups are located on the same side, while in trans isomers, they are located on opposite sides. These isomers may exhibit different physical properties, stability, and biological activities, making them very important in organic and pharmaceutical chemistry.





