Evaporation
Evaporation is one of the most fundamental and extensively applied unit operations in pharmaceutical engineering and industrial pharmacy. It refers to the process of removing a solvent from a solution by converting the liquid solvent into vapor, usually by the application of heat. In pharmaceutical practice, the solvent is most commonly water, although organic solvents such as ethanol, methanol, chloroform, acetone, and isopropyl alcohol are also frequently used depending upon the formulation or extraction process.
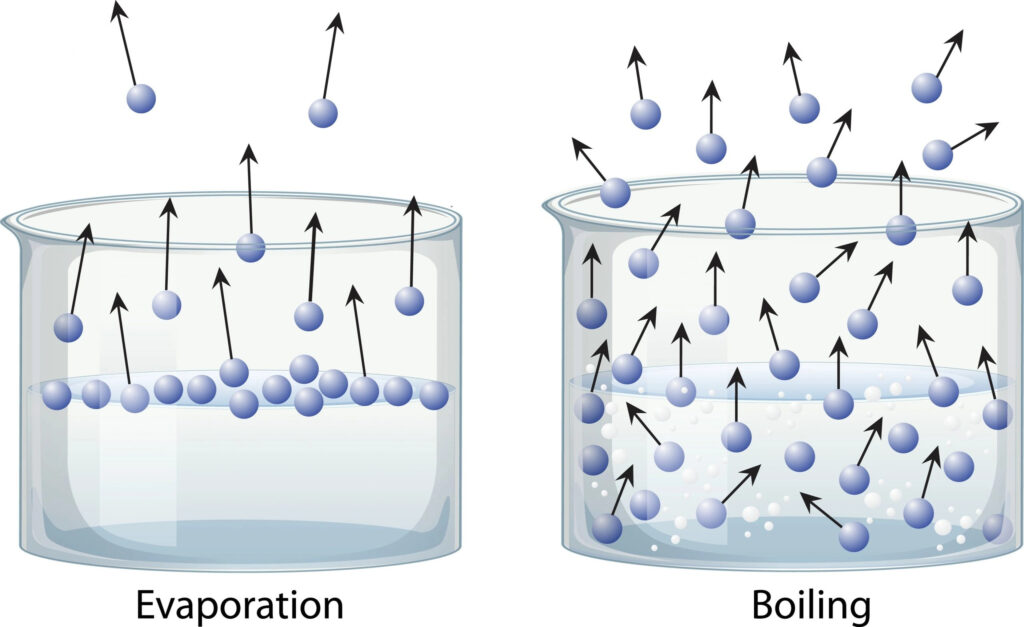
From a scientific standpoint, evaporation is a mass transfer process accompanied by heat transfer, in which molecules at the surface of a liquid gain sufficient energy to escape into the vapor phase. In industrial systems, evaporation is typically carried out under controlled heating conditions to concentrate a solution by removing part of the solvent.
It is important to distinguish evaporation from distillation:
- Evaporation focuses on concentrating the solute by removing solvent.
- Distillation focuses on separating components based on differences in boiling points, often collecting both vapor and residue as valuable products.
In pharmaceutical industries, evaporation is indispensable in the preparation of extracts, syrups, injectables, fermentation products, semi-solid formulations, and solvent recovery systems.
Objectives of Evaporation in Pharmacy
The process of evaporation serves several important objectives in pharmaceutical manufacturing and laboratory operations.
1. Concentration of Pharmaceutical Solutions
The primary objective of evaporation is to increase the concentration of a solution by removing a portion of the solvent.
For example:
- Concentration of herbal extracts after maceration or percolation.
- Preparation of thick syrups by reducing water content.
- Concentration of antibiotic fermentation broths.
By reducing solvent volume, the proportion of active pharmaceutical ingredient (API) per unit volume increases, making the preparation more potent and suitable for further processing.
2. Volume Reduction for Storage and Transportation
Large volumes of dilute pharmaceutical solutions are expensive to store and transport. Evaporation reduces bulk volume, making handling more economical and practical.
For example:
- Bulk drug intermediates may be concentrated before shipment.
- Fermentation broths are concentrated before purification.
3. Solvent Recovery
In pharmaceutical manufacturing, especially during extraction or synthesis, organic solvents are used in large quantities. Evaporation systems often include condensers to recover and reuse solvents.
Advantages:
- Reduces production cost.
- Minimizes environmental pollution.
- Complies with regulatory guidelines for solvent management.
4. Preparation of Semi-Solid and Solid Products
Evaporation is often a preliminary step in:
- Preparation of thick extracts.
- Manufacture of ointments and gels.
- Production of dry powders before spray drying or vacuum drying.
5. Removal of Residual Solvents
After crystallization, synthesis, or granulation, residual solvents may remain. Controlled evaporation ensures compliance with pharmacopeial limits for residual solvents.
6. Improvement of Stability and Shelf Life
Water supports microbial growth and hydrolytic degradation. By reducing water content:
- Microbial contamination decreases.
- Chemical stability improves.
- Shelf life increases.
Thus, evaporation contributes significantly to pharmaceutical quality assurance.
Theoretical Principles of Evaporation
Evaporation involves the interplay of thermodynamics and transport phenomena.
1. Heat Transfer Mechanism
Heat must be supplied to:
- Raise the temperature of the liquid to its boiling point.
- Provide latent heat of vaporization.
The rate of heat transfer depends upon:
- Temperature difference between heating medium and liquid.
- Surface area available for heat exchange.
- Overall heat transfer coefficient.
2. Mass Transfer Mechanism
As solvent molecules escape into vapor form, they must be continuously removed from the surface; otherwise, saturation occurs and evaporation slows down.
3. Vapor Pressure Concept
A liquid boils when its vapor pressure equals atmospheric pressure. If pressure is reduced (as in vacuum evaporation), the boiling point decreases.
This principle is especially important in pharmacy because many drugs are heat-sensitive.
4. Boiling Point Elevation
When a non-volatile solute is dissolved in a solvent:
- The boiling point of the solution increases.
- More energy is required for evaporation.
- Evaporation becomes progressively slower as concentration increases.
This phenomenon must be considered during industrial design.
Applications of Evaporation in Pharmaceutical Industry
Evaporation is widely used across different pharmaceutical domains.
1. Herbal Drug Extraction
In pharmacognosy:
- Plant materials are extracted with solvents.
- The extract is concentrated using evaporation.
- Thick extracts are further processed into tablets, capsules, or syrups.
2. Syrup Manufacturing
Pharmaceutical syrups require:
- Specific sugar concentration (usually 66–67% sucrose).
- Proper viscosity.
- Preservative effect due to high osmotic pressure.
Evaporation ensures precise concentration.
3. Biopharmaceutical Manufacturing
Fermentation processes produce dilute solutions of antibiotics, enzymes, or vaccines. Evaporation:
- Reduces water content.
- Concentrates active biomolecules.
- Prepares solutions for purification or drying.
4. Injectable Formulations
Certain injectables require concentration adjustment before sterilization or lyophilization.
5. Ointment and Gel Preparation
Water removal by evaporation helps achieve:
- Desired consistency.
- Appropriate rheological properties.
6. Solvent Recovery Units
In large-scale pharmaceutical plants:
- Falling film evaporators
- Multiple-effect evaporators
are used to maximize energy efficiency and solvent reuse.
Factors Influencing Evaporation
Evaporation rate and efficiency depend on numerous physical and operational factors.
1. Temperature
Higher temperature:
- Increases kinetic energy of molecules.
- Enhances vapor pressure.
- Accelerates evaporation.
However:
- Excess heat may cause degradation.
- Sensitive drugs require controlled heating.
2. Pressure
Reduced pressure:
- Lowers boiling point.
- Allows evaporation at lower temperatures.
Vacuum evaporation is crucial for:
- Vitamins
- Hormones
- Proteins
- Antibiotics
3. Surface Area
Larger exposed surface area:
- Promotes faster evaporation.
- Improves heat transfer.
Thin film evaporators are designed based on this principle.
4. Nature of Solvent
Volatility plays a key role:
- Ether evaporates rapidly.
- Water evaporates moderately.
- Glycerin evaporates slowly.
5. Concentration of Solute
As solution becomes concentrated:
- Boiling point rises.
- Viscosity increases.
- Heat transfer efficiency decreases.
6. Viscosity
High viscosity:
- Reduces fluid movement.
- Limits convection currents.
- Decreases evaporation rate.
Example: Thick syrups and polymer solutions.
7. Agitation
Stirring:
- Prevents localized overheating.
- Maintains uniform concentration.
- Enhances evaporation rate.
8. Foaming Tendency
Protein-containing solutions may foam:
- Reducing effective evaporation surface.
- Causing product loss.
Antifoaming agents are often used.
9. Scaling and Fouling
Deposition of solids on heating surfaces:
- Reduces heat transfer coefficient.
- Increases energy consumption.
- Requires periodic cleaning.
10. Heat Transfer Coefficient
Depends on:
- Material of evaporator (stainless steel preferred).
- Thickness of heating surface.
- Cleanliness and maintenance.
Advantages of Evaporation
- Simple operation.
- Suitable for large-scale production.
- Energy-efficient when multiple-effect systems are used.
- Improves product stability.
- Allows solvent recovery.
- Adaptable to heat-sensitive drugs (under vacuum).
Limitations of Evaporation
- Risk of thermal degradation.
- Energy-intensive process.
- Not suitable for extremely heat-labile substances without specialized equipment.
- Fouling and maintenance issues.
Pharmaceutical Relevance
For pharmacy students, evaporation bridges:
- Physical Pharmacy (thermodynamics, vapor pressure, boiling point elevation)
- Industrial Pharmacy (equipment design, scale-up)
- Pharmacognosy (extract concentration)
- Biotechnology (fermentation broth concentration)
- Quality Control (residual solvent removal)
A deep understanding of evaporation allows future pharmacists to:
- Optimize formulation processes.
- Prevent drug degradation.
- Improve manufacturing efficiency.
- Ensure regulatory compliance.
Conclusion
Evaporation is a cornerstone unit operation in pharmaceutical science. It serves essential objectives such as concentration of solutions, solvent recovery, volume reduction, and enhancement of product stability. Its efficiency is governed by physical factors like temperature, pressure, viscosity, surface area, and solution composition.
Modern pharmaceutical industries employ advanced evaporators such as vacuum and multiple-effect systems to ensure:
- Energy efficiency
- Product safety
- Stability of heat-sensitive drugs
- Environmental sustainability
A comprehensive understanding of evaporation is therefore indispensable for pharmacy students aiming to excel in industrial pharmacy, formulation science, and pharmaceutical engineering.


1 thought on “Evaporation: Objective, Application, etc”