Safety Data Generation in Drug Development: Ensuring the safety of pharmaceutical products is a foundational objective in drug development. Drug safety, also known as pharmacovigilance, refers to the science and activities concerned with the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problems. Safety data are generated across a continuum of development phases: from preclinical testing in laboratory and animal models to clinical trials in humans, and finally through post-marketing surveillance once the product reaches the market.
Safety Data Generation in Drug Development
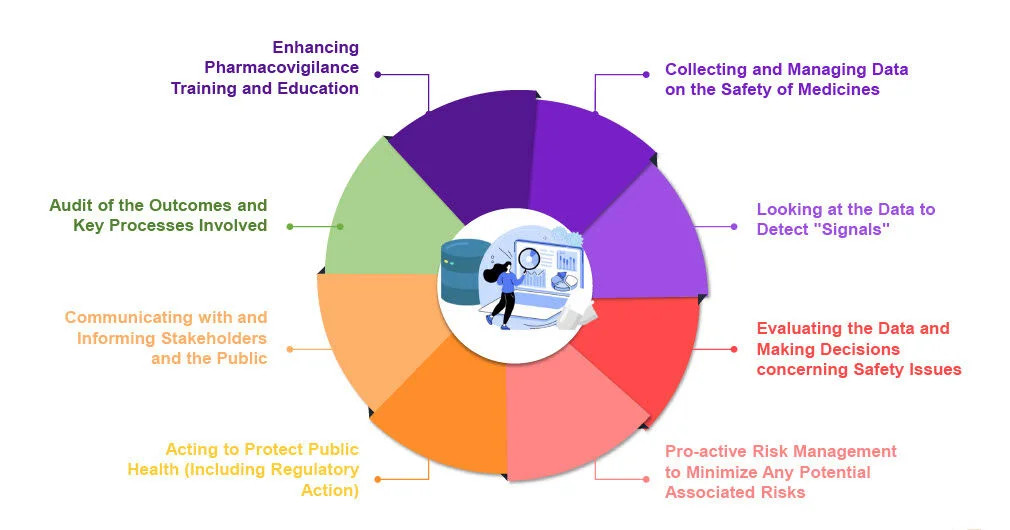
Each of these stages contributes distinct yet interlinked information about a drug’s risk profile. The goal is to build a comprehensive safety dossier that enables informed decisions for prescribers, regulators, and patients.
1. Preclinical Phase: Establishing the Safety Foundation
Before any new drug is tested in humans, it must undergo rigorous preclinical evaluation. This stage is primarily concerned with evaluating basic safety and toxicological profiles in non-human systems, including cell lines and animal models. These investigations are critical for identifying potential hazards, determining safe starting doses for human studies, and fulfilling regulatory requirements for clinical trial approval.
Key Elements of Preclinical Safety Assessment:
A. Pharmacological Profiling
- Primary pharmacodynamics involve studying the intended action of the drug on its target receptor or pathway, which helps confirm its potential therapeutic effect.
- Secondary pharmacodynamics examine unintended biological effects, which could be either beneficial or harmful. Understanding these effects early can predict off-target toxicity or reveal possible repurposing avenues.
B. Toxicology Studies
- Acute Toxicity: Evaluates toxic effects after a single dose. The LD₅₀ (Lethal Dose 50) is calculated to estimate the dose at which 50% of test animals die. This gives an initial impression of the drug’s margin of safety.
- Subacute and Chronic Toxicity: These studies involve repeated administration over weeks to months to understand cumulative toxicity, target organ damage, or delayed effects.
- Genotoxicity: Tests like the Ames test, micronucleus test, and chromosomal aberration assay are used to assess the risk of gene mutation or chromosomal damage, which could lead to cancer.
- Carcinogenicity: Long-term studies (often 18–24 months in rodents) investigate the cancer-causing potential of the compound.
- Reproductive and Developmental Toxicity: Assess the drug’s effect on fertility, embryogenesis, fetal development, and postnatal growth. Teratogenic effects are particularly important for drugs intended for women of childbearing age.
C. Safety Pharmacology
This assesses drug effects on critical physiological systems like:
- Cardiovascular (e.g., QT interval prolongation),
- Respiratory (e.g., respiratory rate and tidal volume), and
- Central Nervous System (e.g., changes in behavior or motor coordination).
D. ADME and Pharmacokinetics
Understanding how the drug is absorbed, distributed, metabolized, and excreted (ADME) informs its bioavailability, accumulation potential, and possible toxic metabolites. These studies help predict systemic exposure in humans and are vital in identifying species-specific responses.
Outcome of Preclinical Phase
The data collectively define a No Observed Adverse Effect Level (NOAEL) and support the Investigational New Drug (IND) application. This package justifies the safety of transitioning the drug into human clinical trials.
2. Clinical Phase: Human Safety and Efficacy Evaluation
The clinical phase involves progressively larger groups of human subjects and is divided into Phase I, II, and III trials. At this stage, safety is no longer hypothetical; it is directly observed in real human biology, where variability, comorbidities, and co-medications are factors.
Phase I: Initial Human Exposure
- Conducted in a small number of healthy volunteers (20–100), unless the drug is highly toxic (e.g., oncology drugs).
- Focus is on safety, tolerability, and pharmacokinetics (how the body affects the drug) and pharmacodynamics (how the drug affects the body).
- Escalating doses are used to determine the maximum tolerated dose (MTD).
- Adverse events (AEs), serious adverse events (SAEs), and vital signs are recorded meticulously.
Example: The disastrous TGN1412 trial in 2006 caused a cytokine storm in volunteers due to an unexpected immune response. This highlighted the importance of understanding immunomodulatory risks even before human testing.
Phase II: Therapeutic Exploration
- Involves a larger cohort (100–300) of patients with the target condition.
- Evaluates efficacy, continues to assess safety, and identifies optimal dosing regimens.
- Reveals common AEs and begins to indicate benefit-risk balance.
- Specific attention is paid to drug interactions, organ-specific toxicities, and early biomarkers of risk.
Example: During Phase II trials of troglitazone, liver enzyme elevations were observed. Though initially deemed manageable, these findings foreshadowed more serious hepatic issues post-marketing.
Phase III: Therapeutic Confirmation
- Conducted on large, diverse populations (1,000–10,000+).
- Confirms therapeutic effectiveness and documents less frequent or late-emerging AEs.
- Provides data for labeling, marketing authorization, and regulatory submission.
- Subgroup analysis (e.g., elderly, pediatric, renal impaired) refines the safety profile for special populations.
Example: Rofecoxib (Vioxx) passed all preclinical and clinical phases, but late Phase III and post-marketing analysis revealed increased cardiovascular risks, leading to its withdrawal.
Safety Monitoring During Clinical Trials
- All adverse events are categorized, coded (e.g., via MedDRA), and analyzed for causality, severity, and expectedness.
- Serious Adverse Events (SAEs) such as death, life-threatening conditions, or hospitalization must be reported within 7–15 days.
- Suspected Unexpected Serious Adverse Reactions (SUSARs) are subject to expedited reporting to regulators and ethics committees.
3. Post-Approval Phase (PMS): Long-Term Real-World Safety
Approval does not mean a drug is fully safe. In fact, many adverse drug reactions (ADRs) only become apparent once the product is used in diverse populations over extended periods. This phase, also known as Post-Marketing Surveillance (PMS), is critical for ongoing risk evaluation.
Sources of Post-Marketing Safety Data:
- Spontaneous Reporting Systems (SRS):
- Voluntary reports by healthcare professionals and consumers.
- Examples include FAERS (USA), EudraVigilance (Europe), and VigiBase (WHO).
- Phase IV (Post-Marketing) Studies:
- Observational or interventional studies to investigate long-term safety, rare events, or special populations (e.g., pregnancy, children).
- Required when regulators impose conditions during approval.
- Active Surveillance:
- Uses electronic health records, insurance claims, or registries to detect trends.
- The FDA Sentinel Initiative is a model system for proactive safety signal detection.
- Real-World Evidence (RWE):
- Incorporates patient data from routine clinical practice to assess safety outside the controlled trial setting.
Signal Detection and Risk Management
- Signal detection involves analyzing large data sets for unexpected associations between a drug and adverse events.
- Once a signal is validated, it may lead to:
- Labeling changes,
- Restrictions on use,
- Medication guides,
- Black box warnings, or
- Market withdrawal in severe cases.
Example: Pioglitazone was linked to bladder cancer in post-marketing data, leading to updated warnings.
Regulatory Reporting Requirements
- Periodic Safety Update Reports (PSURs) or Periodic Benefit-Risk Evaluation Reports (PBRERs) must be submitted at defined intervals.
- Risk Management Plans (RMPs) and Risk Evaluation and Mitigation Strategies (REMS) are tools to minimize known risks while enabling patient access.
Drug Withdrawals Due to Safety Issues
- Thalidomide: Withdrawn after being linked to birth defects in the 1960s, triggering the birth of modern pharmacovigilance.
- Cerivastatin (Baycol): Withdrawn after cases of fatal rhabdomyolysis emerged post-approval.
- Rofecoxib (Vioxx): Withdrawn after cardiovascular risk was confirmed post-marketing.
Conclusion
Safety data generation is not a static process confined to a single phase of drug development—it is an ongoing, evolving commitment that spans from discovery through the entire life cycle of a drug.
- The preclinical phase provides critical early warnings and lays the scientific groundwork for human testing.
- The clinical phase generates structured, high-quality safety data under controlled conditions.
- The post-approval phase captures the drug’s true safety profile in real-world use, often revealing rare, long-term, or population-specific adverse effects.
Robust safety data generation ensures patient safety, builds public trust, and informs evidence-based regulatory decisions. As our tools for data collection and analysis evolve—from electronic health records to artificial intelligence—so too does our ability to proactively manage risks and uphold the integrity of modern medicine.

