Aim: Preparation of Calcium Gluconate Injection
Objective
The primary objective of this experiment is to prepare a sterile, parenteral formulation of calcium gluconate, a biologically active calcium salt, suitable for intravenous administration. The experiment also aims to demonstrate accurate solution preparation, pH adjustment, sterilization techniques, and adherence to pharmaceutical quality control standards.
Introduction
Calcium gluconate is the calcium salt of gluconic acid, widely employed in the medical field to correct hypocalcemia (low blood calcium levels), hyperkalemia (high potassium levels), magnesium toxicity, and as a cardioprotective agent during certain conditions. Calcium is a vital cation involved in multiple physiological processes, including blood coagulation, nerve conduction, muscle contraction, and enzymatic regulation.
Chemical Profile of Calcium Gluconate:
- Chemical Name: Calcium D-gluconate
- Molecular Formula: C₁₂H₂₂CaO₁₄
- Molecular Weight: 430.37 g/mol (anhydrous)
- Appearance: White crystalline powder
- Solubility: Freely soluble in water; practically insoluble in ethanol
- Pharmacological Use: Treatment of calcium deficiency, hypocalcemia, hyperkalemia, and magnesium sulfate overdose.
Significance of Parenteral Preparation:
Oral calcium supplements may have limited absorption, delayed onset, or gastrointestinal irritation. Parenteral calcium gluconate provides rapid correction of calcium deficits, crucial in emergency conditions such as acute hypocalcemia, tetany, and cardiac arrhythmias.
Materials and Equipment
Materials
- Calcium gluconate (anhydrous or monohydrate)
- Sterile distilled water for injection
- pH-adjusting agents: dilute hydrochloric acid (HCl) or sodium hydroxide (NaOH)
- Preservatives (optional, e.g., benzyl alcohol 0.9% w/v)
Equipment
- Analytical balance (accuracy ±0.1 mg)
- Magnetic stirrer with heating (if required)
- Glass beakers, volumetric flasks, graduated cylinders
- pH meter or high-precision pH paper
- Filtration assembly with sterilizing-grade 0.22 μm membrane
- Autoclave (121°C, 15 psi, 15–20 min)
- Laminar airflow chamber for aseptic manipulations
- Ampoules or sterile vials for final product
- Hot air oven (for drying ampoules, if needed)
Formulation Example (100 mL Injection)
| Ingredient | Quantity | Function |
| Calcium gluconate | 9.0 g | Active pharmaceutical ingredient |
| Sterile distilled water | q.s. to 100 mL | Vehicle / solvent |
| Hydrochloric acid / NaOH | q.s. to pH 6.5–8.0 | pH adjustment |
| Benzyl alcohol (optional) | 0.9 g | Preservative (if multi-dose) |
Remarks:
- A 10% w/v solution is standard for intravenous administration.
- pH adjustment is crucial to avoid phlebitis and vein irritation.
Theoretical Calculations
Step 1: Determine weight of calcium gluconate required
- Target concentration: 10% w/v
Amount required = 10g per 100 mL solution
Step 2: Adjust for monohydrate (if using CaC₁₂H₂₂O₁₄·H₂O)
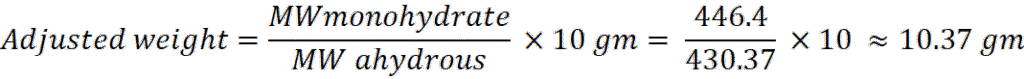
Step 3: pH adjustment
- Use dilute HCl to lower pH if >8.0
- Use dilute NaOH to increase pH if <6.5
- Final pH: 6.5–8.0
Procedure
Preparation of Aqueous Solution
- Accurately weigh the required amount of calcium gluconate.
- Dissolve the salt in approximately 80% of the final volume of sterile distilled water using a magnetic stirrer for uniform dissolution.
- Measure pH using a calibrated pH meter; adjust to 6.5–8.0 using dilute HCl or NaOH.
- Make up the solution to the final volume with sterile distilled water.
Sterilization of Solution
- Filtration method (preferred for heat-sensitive solutions): Pass the solution through a 0.22 μm sterilizing filter into a sterile receiving vessel under aseptic conditions.
- Autoclaving (if solution is heat-stable): Fill sterile vials or ampoules and autoclave at 121°C for 15 min at 15 psi.
Filling and Sealing
- Fill sterile solution into pre-sterilized ampoules or vials under a laminar airflow hood.
- Seal ampoules by flame or cap vials with sterile rubber stoppers and aluminum seals.
- Label with drug name, concentration, batch number, and expiry date.
Quality Control Tests
- Appearance: Should be clear, colorless, and free from particulate matter.
- pH Measurement: 6.5–8.0
- Sterility Test: Perform as per USP <71> Sterility Tests
- Assay of Calcium Content: Can be performed via complexometric titration with EDTA or spectrophotometric methods.
- Pyrogen Test: Ensure endotoxin-free via rabbit pyrogen test or LAL assay.
- Particulate Matter: Must comply with USP <788> guidelines for injectable solutions.
Storage Conditions
- Store in tight, light-resistant containers.
- Maintain room temperature (15–25°C).
- Protect from light, freezing, and contamination.
- Single-dose ampoules should be used immediately; multi-dose vials must include a preservative and be handled under aseptic conditions.
Precautions and Safety Measures
- Ensure strict aseptic technique throughout preparation to prevent microbial contamination.
- Always confirm pH to prevent venous irritation upon injection.
- Avoid overheating if autoclaving, as calcium gluconate may decompose at excessive temperatures.
- Dispose of any contaminated solutions or broken ampoules according to lab safety protocols.
- Verify the identity, concentration, and sterility before administration.
References
- United States Pharmacopeia (USP 43-NF 38), Calcium Gluconate Injection Monograph.
- Indian Pharmacopoeia (IP), 2020, Volume II.