1. Introduction of Kinetics of Elimination
Drug elimination is a vital pharmacokinetic process that refers to the irreversible removal of active drug molecules from the body. It primarily occurs through metabolism (biotransformation) and excretion. Together with absorption and distribution, elimination determines the duration and intensity of a drug’s pharmacological action.
Understanding the kinetics of elimination is essential in clinical pharmacology because it affects dosing regimens, drug accumulation, therapeutic efficacy, and the risk of toxicity. Elimination kinetics helps define how long a drug remains in the systemic circulation and how often it should be administered to maintain desired plasma levels.
2. Basic Definitions
Elimination Rate: The rate at which a drug is cleared from the body, typically expressed in mg/hour or % per hour.
Clearance (Cl): The volume of plasma completely cleared of drug per unit time.
Half-life (t₁/₂)
Time required for the plasma concentration of a drug to reduce by half.
Volume of Distribution (Vd)
Theoretical volume that would be required to contain the total amount of drug in the body at the same concentration as in plasma.
3. Types of Elimination Kinetics
There are two primary types of elimination kinetics: First-order kinetics and Zero-order kinetics, with Mixed-order (nonlinear) kinetics occurring in some cases.
A. First-Order Kinetics (Exponential Elimination)
Definition: In first-order kinetics, the rate of drug elimination is directly proportional to the drug concentration in plasma. This means that a constant fraction or percentage of drug is eliminated per unit of time, regardless of the dose.
- In simple terms: The more drug there is in the body, the faster it gets eliminated.
- However, only a constant percentage of the drug is eliminated per unit time, not a constant amount.
Example:
Let’s say 10% of a drug is eliminated per hour:
- If the plasma concentration is 100 mg → 10 mg eliminated in 1 hour.
- If the concentration is 50 mg → 5 mg eliminated in 1 hour.
Characteristics
- Most drugs follow first-order elimination: This is the most common type of elimination for drugs at therapeutic doses.
Examples: Paracetamol, Atenolol, and Theophylline.
- Plasma concentration decreases exponentially over time: The concentration-time curve is exponential, not linear.
It means the drug concentration halves in regular time intervals (see half-life below).
- Half-life is constant and independent of dose: Half-life is the time it takes for the drug’s plasma concentration to reduce by 50%. In first-order kinetics, the half-life remains unchanged, no matter the starting dose.
- Clearance remains constant: Clearance (CL) refers to the volume of plasma from which the drug is completely removed per unit time. In first-order kinetics, the body clears the drug at a constant rate based on concentration.
Graphical Representation
- On a linear scale: Curved decay
- On a semi-logarithmic plot: Straight line
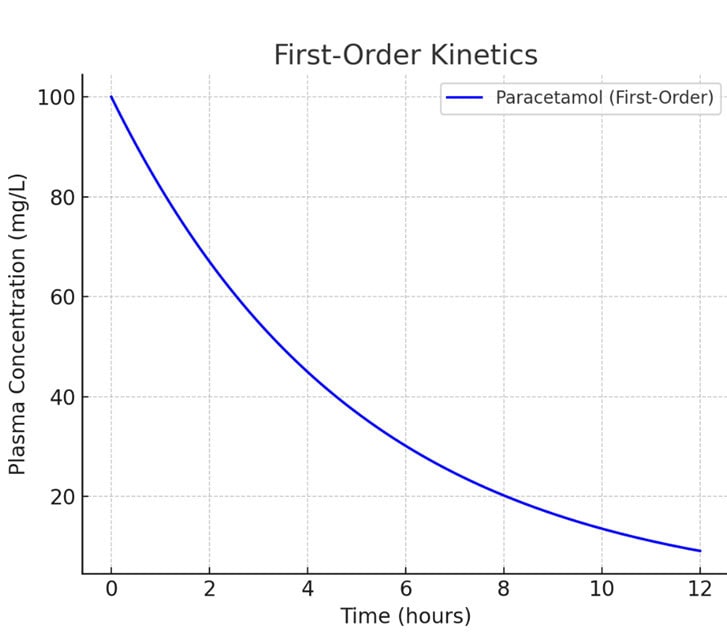
First-Order Kinetics (Paracetamol)
- Graph Insight: Exponential decline in plasma concentration over time.
- Mechanism: A constant fraction of the drug is eliminated per unit time.
- Equation: C = C0 ⋅ e−kt
- Example: Most drugs, like Paracetamol, follow this pattern.
Equation
C = C0 ⋅ e−kt
Where:
- C = concentration at time t
- C₀ = initial concentration
- k = elimination rate constant
Clinical Implications
- Easier to predict drug accumulation and design dosing intervals.
- Most drugs like penicillin, paracetamol, theophylline, propranolol, and amoxicillin follow this pattern.
B. Zero-Order Kinetics (Linear Elimination)
Definition: In zero-order kinetics, the rate of elimination is constant, independent of the drug concentration. A fixed amount of drug is eliminated per unit time.
Unlike first-order kinetics (where a constant percentage is removed), here a constant quantity is removed.
Example:
If the elimination rate is 10 mg/hour:
- 100 mg in plasma → 10 mg eliminated in 1 hour.
- 80 mg in plasma → still 10 mg eliminated in 1 hour.
So, the amount removed stays the same, even if drug levels are high or low.
Characteristics
- Occurs when eliminating enzymes are saturated: The enzymes responsible for metabolism or elimination (e.g., in the liver) become fully saturated. They can’t speed up elimination even if the drug concentration increases. This typically happens at high doses of some drugs.
- Plasma concentration decreases linearly over time: Since a fixed amount is removed every hour, the concentration drops in a straight line on a graph (not exponential).
- Half-life is not constant—it increases with dose: Half-life varies because it takes longer to eliminate half the drug at higher concentrations.
Example: If 10 mg/hr is eliminated, it takes more time to remove half of 100 mg than half of 50 mg.
- Clearance is not constant—it decreases as dose increases: Clearance depends on how fast the drug is eliminated relative to its concentration. Since elimination rate doesn’t increasewith dose, but concentration does, clearance drops as the dose rises.
Graphical Representation
- On a linear scale: Straight line
- On a semi-logarithmic scale: Curved line
Zero-Order Kinetics (Ethanol)
- Graph Insight: Linear decline in plasma concentration.
- Mechanism: A constant amount of drug is eliminated per unit time, independent of concentration.
- Equation: C = C0 – k ⋅ t
- Example: Ethanol, Phenytoin (at high doses), and Aspirin (in toxic doses).
Examples of Drugs
- Phenytoin
- Ethanol (alcohol)
- Aspirin (in high doses)
Clinical Implications
- Small increases in dose may lead to disproportionately high plasma levels.
- Higher risk of toxicity and overdose.
- Dosing must be done with extreme care, and therapeutic drug monitoring (TDM) is essential.
C. Mixed-Order Kinetics / Michaelis-Menten Kinetics
Definition: Some drugs exhibit dose-dependent elimination kinetics, where they follow first-order kinetics at low concentrations and zero-order kinetics at high concentrations due to enzyme saturation.
- At low concentrations, the drug follows first-order kinetics (a constant fraction is eliminated per unit time).
- At high concentrations, the drug follows zero-order kinetics (a constant amount is eliminated per unit time).
Mixed-Order or Michaelis-Menten Kinetics (Phenytoin)
- Graph Insight: Initially shows first-order behavior, then plateaus as elimination capacity saturates.
- Mechanism: Follows first-order at low concentrations; becomes zero-order at higher concentrations when enzymes are saturated.
- Equation: Rate =
- Example: Phenytoin, Theophylline, and Warfarin under certain conditions.
Why It Happens:
- The body eliminates many drugs using enzymes (e.g., in the liver).
- At low doses, these enzymes work well below their maximum capacity—so the rate depends on how much drug is present (first-order).
- At high doses, enzymes get saturated, and cannot work faster—so the elimination becomes constant, regardless of concentration (zero-order).
Examples
- Phenytoin
- Theophylline
- Warfarin
Michaelis-Menten Equation
Where:
- v = rate of drug metabolism
- Vₘₐₓ = maximum rate of metabolism
- Kₘ = Michaelis constant (drug concentration at which v = ½ Vₘₐₓ)
Clinical Implications
- Difficult to predict plasma levels.
- Requires careful titration and individualized dosing.
4. Pharmacokinetic Parameters Related to Elimination
| Parameter | Symbol | Description |
| Elimination rate constant | k | Fraction of drug eliminated per unit time |
| Half-life | t₁/₂ | Time taken to reduce plasma concentration by half |
| Clearance | Cl | Volume of plasma cleared of drug per unit time |
| AUC (Area Under Curve) | – | Total drug exposure over time |
| Steady-state concentration | C_ss | Plasma concentration when rate of administration equals rate of elimination |
5. Elimination and Steady-State
When a drug is administered repeatedly (e.g., in chronic therapy), it accumulates in the body until a steady-state concentration (Css) is achieved, where input equals output.
Time to Steady-State
- Takes about 4 to 5 half-lives to reach steady-state levels in first-order kinetics.
- Important for chronic therapy, such as in antibiotics, antiepileptics, or cardiac drugs.
6. Factors Influencing Elimination Kinetics
- Age – Neonates and elderly have reduced enzyme activity.
- Renal/Hepatic function – Impaired kidney or liver affects elimination.
- Genetic polymorphisms – Enzyme variants can speed up or slow down metabolism.
- Drug interactions – Enzyme inhibitors or inducers alter elimination kinetics.
- Disease states – Conditions like liver cirrhosis or heart failure can reduce clearance.
- Route of administration – Affects bioavailability and metabolism pathway.
7. Clinical Applications
- Dose adjustment in renal or hepatic impairment based on clearance.
- Therapeutic drug monitoring (TDM) for drugs with narrow therapeutic index.
- Avoiding toxicity in drugs with zero-order kinetics.
- Optimizing dosage intervals to maintain effective drug levels.